Pudendal Neuralgia
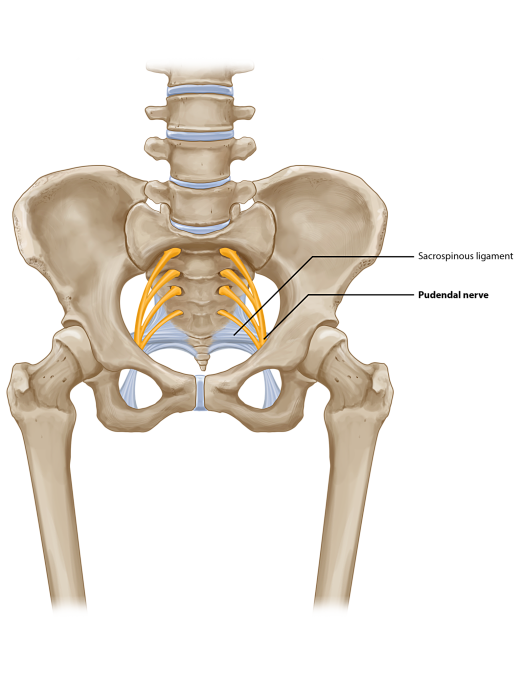
Pudendal neuralgia (PN) is defined as the existence of pain in the distribution of the pudendal nerve. Pudendal neuropathy can occur in both genders. Entrapment of the pudendal nerve can give rise to perineal pain involving the penis, scrotum, labia, perineum, or anorectal region. Three sites of entrapment of the nerve have been described. These can occur along its path between the sacrotuberous and the sacrospinous ligaments (interligamentous plane), through Alcock canal, or as the nerve passes through a tight osteofibrotic canal at the entrance to the base of the penis.
Synonyms include pudendal nerve entrapment or the syndrome of the pudendal canal (Alcock canal). Clinicians rarely associate these symptoms with pudendal neuropathy. Conventional diagnoses include “prostatitis” or “prostadynia” in males or endometriosis or vulvodynia in females.