Chronic Pelvic Pain Syndrome in Women: Advanced Pain Management and Interventional Treatments

Chronic Pelvic Pain Syndrome in Women
Pain Spa | Dr M. Krishna | Advanced Pain Management • Interventional Treatments • Pelvic Floor Therapy
The Myofascial Component of CPPS: Why It Develops and Why It Matters
Myofascial pain is one of the most common yet frequently overlooked contributors to chronic pelvic pain syndrome (CPPS). Depending on the population studied and the examination techniques used, myofascial dysfunction is identified in approximately 22% to 94% of women with chronic pelvic pain. In specialist pelvic pain clinics that routinely perform a detailed musculoskeletal assessment, 50% to 90% of women are found to have pain arising from muscles, fascia, and related connective tissues.
This is clinically important because myofascial pain often persists even after treatment of the original gynecological condition. A woman may undergo successful excision of endometriosis, hormonal suppression, hysterectomy, or bladder-directed treatment yet continue to experience pelvic pain because the pelvic floor and surrounding muscles have become independent pain generators. In many patients, untreated myofascial dysfunction is the principal reason why organ-specific treatments fail to provide complete relief.
Key Clinical Message: Persistent pelvic pain does not necessarily mean that the original disease is still active. In many women, the muscles and nerves surrounding the pelvis continue to generate pain long after the initial trigger has been treated.
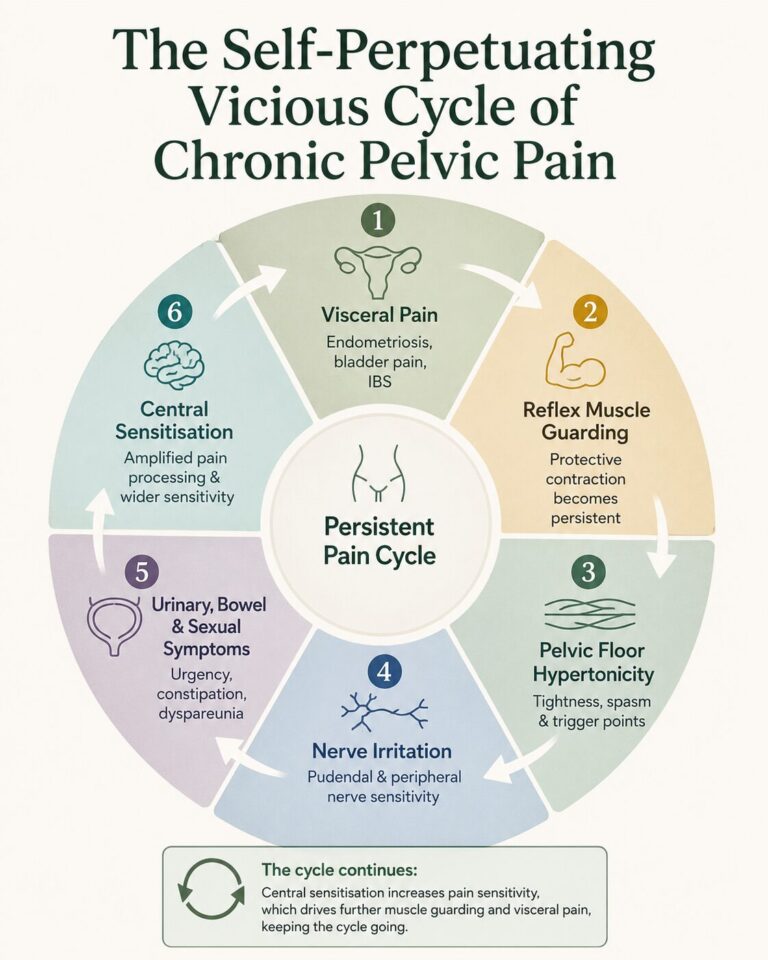
The development of myofascial pain is best understood through the concept of viscerosomatic convergence. Visceral structures such as the uterus, bladder, and bowel share spinal cord pathways with somatic structures including muscles, fascia, ligaments, and peripheral nerves. Persistent nociceptive input from endometriosis, bladder pain syndrome, irritable bowel syndrome, or other visceral conditions can trigger reflex contraction and guarding of the pelvic floor and surrounding muscles.
A related phenomenon, viscero-viscero cross-sensitisation, explains why multiple pelvic conditions frequently coexist. Activity in one organ can increase sensitivity in another. For example, endometriosis may sensitise the bladder and bowel, while bladder pain syndrome may amplify uterine or rectal pain.
Sustained muscle contraction reduces local blood flow, creating an “energy crisis” within the muscle. This leads to trigger point formation, release of inflammatory mediators, and increasing sensitivity of both muscle and nerve tissue. Over time, the muscles themselves become independent generators of pain.
The process then becomes bidirectional. Hypertonic pelvic muscles can compress the urethra, bladder base, rectum, and pudendal nerve, causing urinary urgency, incomplete emptying, constipation, and sexual pain. Persistent input from these muscles also drives central sensitisation, resulting in widespread pain, sleep disturbance, fatigue, and heightened sensitivity.
A landmark study by Stratton and colleagues demonstrated that signs of sensitisation were present in more than 80% of women with chronic pelvic pain, and myofascial trigger points were identified in over 90%. Women with trigger points were significantly more likely to exhibit allodynia and hyperalgesia.
The practical implication is simple: successful treatment of CPPS requires addressing not only the original pelvic pathology but also the muscular, neural, visceral, and central nervous system mechanisms that sustain pain.
High-Tone Pelvic Floor Dysfunction: Pathogenesis, Clinical Features and Detailed Assessment
High-tone pelvic floor dysfunction is one of the most important myofascial pain generators in chronic pelvic pain syndrome. It describes a state in which the pelvic floor muscles remain excessively contracted, tender and difficult to relax. This may begin as a protective response to pelvic pain, but over time it can become an independent source of pain, urinary symptoms, bowel symptoms and sexual dysfunction.
In the context of CPPS, pelvic floor hypertonicity is often reactive rather than primary. Visceral pain from conditions such as endometriosis, bladder pain syndrome or irritable bowel syndrome can trigger reflex pelvic floor guarding through shared spinal cord pathways. Once this guarding becomes persistent, the muscles develop reduced blood flow, trigger points, increased sensitivity and impaired relaxation.
Key Clinical Message: High-tone pelvic floor dysfunction is not simply “tight muscles”. It is a pain-processing disorder involving muscle guarding, trigger points, nerve irritation, visceral sensitivity and central sensitisation.
Several mechanisms can contribute to pelvic floor hypertonicity. Reflex guarding may occur in response to ongoing visceral pain. Trauma-related hypervigilance may keep the pelvic floor in a protective contracted state. Postsurgical changes, scarring, mesh procedures, childbirth-related injury and prolonged pain after pelvic operations may also alter pelvic floor tone and nerve sensitivity.
The pudendal nerve also plays an important role. It carries sensory and motor fibres from the sacral nerve roots and contributes to pelvic floor motor control, perineal sensation, sphincter function and sexual function. Persistent pelvic floor spasm can irritate the pudendal nerve, while pudendal nerve sensitivity can further increase pelvic floor guarding. This creates a feedback loop between the nerve, muscles and spinal cord.
Typical Clinical Features
| Pain pattern | Persistent pelvic heaviness, deep aching, pressure, burning or pain radiating to the groin, vagina, rectum, buttock or legs. |
| Sexual symptoms | Deep dyspareunia, pain during intercourse, pain continuing after intercourse and avoidance of intercourse because of anticipated flares. |
| Urinary symptoms | Urgency, frequency, incomplete emptying, bladder pressure or a persistent sensation of needing to pass urine despite low bladder volume. |
| Bowel symptoms | Constipation, difficult evacuation, rectal pressure, pain with defaecation or a sensation of incomplete bowel emptying. |
| Activity-related features | Pain worsened by walking, exercise, prolonged sitting, cycling, pelvic loading or stress-related muscle guarding. |
A detailed pelvic muscle assessment is therefore essential. The aim is not simply to identify tenderness, but to determine whether palpation reproduces the patient’s familiar pain. This distinction is important because many patients have general discomfort during examination, but clinically meaningful myofascial pain is suggested when pressure over a specific muscle recreates the symptoms they experience in daily life.
A data-driven assessment approach involves palpation of six pelvic muscle sites: the right and left pubococcygeus, right and left iliococcygeus, and right and left obturator internus. Each site can be scored from 0 to 10 for tenderness. A total tenderness score of 12 or more out of 60 has been shown to have good diagnostic accuracy for high-tone pelvic floor dysfunction.
Pelvic Muscle Assessment: Practical Interpretation
| Assessment Finding | Clinical Meaning |
|---|---|
| High tenderness score | Suggests significant pelvic floor myofascial involvement. |
| Reproduction of familiar pain | Supports that the muscle being examined is a clinically relevant pain generator. |
| Difficulty relaxing the pelvic floor | Suggests impaired down-regulation and persistent guarding. |
| Obturator internus tenderness | May indicate a deep pelvic and hip-rotator contribution to pelvic, groin, vaginal or rectal pain. |
| Associated urinary or bowel symptoms | May reflect somatovisceral effects of hypertonic pelvic muscles on bladder and bowel function. |
The examination should be interpreted functionally. For example, tenderness in the levator ani may help explain deep dyspareunia and rectal pressure, while obturator internus tenderness may contribute to pelvic sidewall pain, hip-related pain and pain worsened by sitting or activity. The goal is to map the patient’s symptoms to the relevant muscular, neural and visceral contributors.
This assessment guides treatment. Patients with high-tone pelvic floor dysfunction usually require pelvic floor down-training, myofascial release, treatment of visceral drivers, and where appropriate, image-guided trigger point injections, botulinum toxin, pudendal nerve blocks or neuromodulation strategies. Treating the pelvic floor in isolation is often insufficient; the most effective approach addresses the muscle, nerve, visceral and central pain mechanisms together.
Major Myofascial Pain Generators in Chronic Pelvic Pain Syndrome
Chronic pelvic pain syndrome is rarely caused by one isolated muscle. In many women, several anterior, pelvic floor, hip and deep gluteal muscles become involved together. These muscles may act as primary pain generators, secondary guarding responses to visceral pain, or contributors to nerve irritation and central sensitisation.
A structured musculoskeletal assessment is therefore essential. The examination should include the anterior abdominal wall, pelvic floor, obturator internus, deep gluteal muscles, iliopsoas, hip adductors, pubic symphysis and sacroiliac region. The aim is to identify which muscles reproduce the patient’s familiar pain and which structures are maintaining the wider pain cycle.
Key Clinical Message: The pelvic floor should not be assessed in isolation. Abdominal wall trigger points, hip flexor dysfunction, obturator internus spasm, piriformis-related pain and adductor-pubic symphysis problems may all perpetuate chronic pelvic pain.
Anterior Abdominal Wall
The anterior abdominal wall is a common but often missed contributor to CPPS. Trigger points in the rectus abdominis, internal oblique, external oblique and transversus abdominis may produce lower abdominal or pelvic pain that mimics visceral pathology. Scar-related trigger points after caesarean section, laparoscopy or abdominal surgery may also contribute to persistent pain.
The Carnett test is a key bedside assessment. If the patient’s pain remains the same or worsens when the abdominal muscles are tensed, this supports an abdominal wall source rather than a purely visceral cause. Anterior cutaneous nerve entrapment syndrome (ACNES) should be considered when pain is focal, located near the lateral border of the rectus muscle, and associated with sensory change or a positive pinch test.
Pelvic Floor Muscles
The levator ani complex is one of the most frequently involved muscle groups in CPPS. This includes the pubococcygeus, puborectalis and iliococcygeus muscles. Tenderness, spasm or impaired relaxation in these muscles may contribute to deep pelvic pain, dyspareunia, rectal pressure, urinary urgency, pelvic heaviness and pain flares after intercourse or activity.
The coccygeus muscle may contribute to posterior pelvic pain, rectal discomfort and coccydynia-type symptoms. Internal pelvic floor assessment should evaluate tone, tenderness, trigger points and the patient’s ability to voluntarily contract and relax the pelvic floor.
Obturator Internus
The obturator internus is particularly important because it lies along the lateral pelvic wall and has close anatomical relationships with the bladder, urethra, vagina, rectum and pudendal nerve. It may contribute to pelvic sidewall pain, deep vaginal or rectal pain, hip and buttock pain, dyspareunia, urinary symptoms and pain worsened by sitting.
Assessment may include transvaginal palpation of the anterolateral pelvic wall, resisted external hip rotation and hip provocation tests such as FABER. In selected patients, treatment may involve pelvic floor physiotherapy, trigger point injection or image-guided obturator internus injection.
Piriformis and Deep Gluteal Contributors
The piriformis and deep gluteal muscles may contribute to posterior pelvic pain, buttock pain, sitting intolerance and sciatica-like symptoms. The piriformis, gemelli, obturator internus and quadratus femoris form part of a deep hip-rotator system that may interact with the sciatic nerve and pudendal nerve.
When these muscles are tight or painful, they may contribute to deep gluteal syndrome, referred pelvic pain or neural irritation. Assessment should include palpation of the deep gluteal region, hip rotation testing, sitting-related symptom provocation and evaluation for sciatic or pudendal-type symptoms.
Iliopsoas and Iliacus
The iliopsoas and iliacus muscles may contribute to anterior pelvic pain, groin pain, low back pain and pain with hip flexion, climbing stairs, prolonged sitting or walking. These muscles are clinically important because of their relationship with the lumbar plexus, including the genitofemoral and lateral femoral cutaneous nerves.
Genitofemoral irritation may cause burning, tingling or radiating pain into the groin, mons pubis, labia majora or upper medial thigh. Assessment may include the Thomas test, resisted hip flexion, deep abdominal palpation and ultrasound-guided evaluation where appropriate.
Hip Adductors and Pubic Symphysis
The hip adductors and pubic symphysis can contribute to medial groin pain, pubic pain and anterior pelvic pain. The adductor longus, adductor brevis, adductor magnus, pectineus and gracilis may become painful through overuse, altered pelvic mechanics, childbirth-related strain, athletic loading or compensatory guarding.
Pubic symphysis dysfunction, osteitis pubis and athletic pubalgia may overlap with CPPS and should be considered when pain is localised to the pubic region, worsens with resisted adduction, or is associated with groin loading. Assessment may include pubic palpation, adductor squeeze testing, FABER testing and evaluation for coexisting hip pathology.
Major Muscle Groups and Their Clinical Clues
| Muscle / Region | Typical Clues | Assessment / Treatment Direction |
|---|---|---|
| Rectus abdominis / abdominal wall | Focal lower abdominal pain, scar sensitivity, pain with sit-ups or abdominal tension. | Carnett test, pinch test, trigger point injection, ACNES assessment. |
| Obliques / transversus abdominis | Lower quadrant pain, pain with twisting, post-inguinal or abdominal surgery pain. | Palpation lateral to rectus, resisted trunk rotation, ilioinguinal or iliohypogastric nerve assessment. |
| Levator ani complex | Deep pelvic pain, dyspareunia, rectal pressure, urinary urgency or pelvic heaviness. | Internal pelvic floor assessment, down-training, myofascial release, trigger point injection. |
| Coccygeus | Posterior pelvic pain, rectal discomfort, coccyx-related pain. | Posterior pelvic floor palpation, coccyx assessment, pelvic floor therapy. |
| Obturator internus | Pelvic sidewall pain, hip or buttock pain, sitting pain, dyspareunia, urinary symptoms. | Anterolateral vaginal wall palpation, resisted external rotation, image-guided injection if appropriate. |
| Piriformis / deep gluteal muscles | Buttock pain, sitting intolerance, sciatica-like symptoms, pudendal irritation. | Deep gluteal palpation, hip rotation tests, ultrasound-guided piriformis or sciatic-level assessment. |
| Iliopsoas / iliacus | Anterior pelvic pain, groin pain, pain with hip flexion, stairs or prolonged sitting. | Thomas test, resisted hip flexion, genitofemoral nerve consideration, ultrasound-guided treatment. |
| Hip adductors / pubic symphysis | Medial groin pain, pubic pain, pain with resisted adduction or athletic loading. | Adductor squeeze test, pubic palpation, FABER test, targeted rehabilitation or injection. |
The key clinical point is that myofascial CPPS should be mapped by symptom pattern and examination findings rather than by imaging alone. Treatment is most effective when the relevant muscle groups are identified, the patient’s familiar pain is reproduced on examination, and therapy is directed toward the full chain of muscular, neural and visceral contributors.
Posterior Pelvic Pain Generators: Sacroiliac Joint Dysfunction and Coccydynia
Posterior pelvic pain is an important and often overlooked component of chronic pelvic pain syndrome. Two key structures to assess are the sacroiliac joint and the coccyx. Both can produce pain that overlaps with pelvic floor dysfunction, buttock pain, perineal pain and sitting intolerance.
The sacroiliac joint may contribute to pain below the L5 region, unilateral buttock pain, pain during sit-to-stand transitions and postpartum pelvic girdle pain. The Fortin finger test, where the patient points with one finger to the painful area over the posterior superior iliac spine, can be a useful clinical clue. A cluster of provocation tests, including FABER, Gaenslen, compression, distraction and thigh thrust, may support sacroiliac joint involvement.
Key Clinical Message: The sacroiliac joint, pelvic floor and coccyx are biomechanically linked. Sacroiliac dysfunction may drive pelvic floor guarding, pelvic floor spasm may worsen coccygeal pain, and coccygeal pathology may increase levator ani and coccygeus tone.
Diagnostic sacroiliac joint injections can help confirm whether the joint is a significant pain generator. When sacroiliac pain is confirmed and persists despite physiotherapy, rehabilitation and injection-based care, radiofrequency treatment of the sacral lateral branches may be considered. In selected patients, cooled radiofrequency can create a larger treatment zone and may be useful where the pain pattern and diagnostic response support sacroiliac joint-mediated pain.
Coccydynia refers to pain arising from the coccyx or sacrococcygeal joint. It is particularly relevant in pelvic pain because the coccyx acts as a posterior anchor for the pelvic floor muscles, including the coccygeus and levator ani. Coccygeal pain may be triggered by trauma, childbirth, prolonged sitting, cycling, rising from sitting or defaecation.
In women with pelvic pain, coccydynia may coexist with coccygeus spasm, anococcygeal ligament pain, sacrococcygeal joint restriction, impaired pelvic floor coordination and outlet-type constipation. This means that coccyx pain should not be viewed as an isolated tailbone problem; it may be part of a wider posterior pelvic floor pain pattern.
Posterior Pelvic Pain: Clinical Clues and Treatment Direction
| Pain Generator | Typical Clinical Clues | Assessment and Treatment Direction |
|---|---|---|
| Sacroiliac joint dysfunction | Posterior pelvic pain below L5, unilateral buttock pain, pain with sit-to-stand transitions, postpartum pelvic girdle pain, positive Fortin finger test. | Provocation test cluster, diagnostic sacroiliac joint injection, therapeutic injection, sacral lateral branch radiofrequency or cooled radiofrequency in selected cases. |
| Coccyx / sacrococcygeal joint | Pain with prolonged sitting, rising from sitting, cycling or defaecation; history of fall, childbirth trauma or coccygeal instability. | External and internal coccygeal assessment, dynamic imaging where appropriate, pelvic floor therapy, coccygeal mobilisation, sacrococcygeal injection, ganglion impar block. |
| Coccygeus and posterior pelvic floor | Posterior pelvic pain, rectal pressure, coccydynia-type symptoms, pain with bowel opening or pelvic floor spasm. | Posterior vaginal wall palpation, coccygeal mobility assessment, pelvic floor down-training, trigger point injection if appropriate. |
| Ganglion impar-mediated pain | Perineal, coccygeal, distal rectal, vulval or distal vaginal pain with sympathetic or deep aching features. | Image-guided ganglion impar block; pulsed radiofrequency may be considered for persistent non-malignant coccygeal or perineal pain. |
The ganglion impar is an important target in refractory coccygeal and perineal pain. It lies anterior to the sacrococcygeal junction and carries nociceptive and sympathetic input from the perineum, distal rectum, perianal region, distal urethra, vulva and distal vagina. A diagnostic ganglion impar block may help identify whether this pathway is contributing to the patient’s pain.
For persistent coccydynia or perineal pain, pulsed radiofrequency of the ganglion impar may be considered in carefully selected patients. This approach aims to modulate pain signalling without destroying the nerve, and may be preferable to neurolytic procedures for non-malignant pain when longer-lasting benefit is needed.
The practical lesson is that posterior pelvic pain should be assessed as a connected system rather than as separate diagnoses. Sacroiliac dysfunction, coccygeal pain and pelvic floor hypertonicity often reinforce each other, so treatment should combine rehabilitation, pelvic floor therapy, diagnostic blocks and targeted image-guided interventions where appropriate.
Peripheral Neuralgias in Chronic Pelvic Pain: Pudendal Neuralgia and Other Nerve Irritation Syndromes
Peripheral nerve irritation is an important contributor to chronic pelvic pain syndrome. In many patients, the problem is not a fixed anatomical entrapment, but a sensitised or irritated nerve within a wider pain system involving pelvic floor hypertonicity, scar tissue, inflammation, altered movement patterns and central sensitisation.
Pudendal neuralgia is the most recognised pelvic neuralgia, but it should not automatically be assumed to mean pudendal nerve entrapment. The pudendal nerve may become irritated by pelvic floor spasm, obturator internus hypertonicity, prolonged sitting, cycling, childbirth trauma, pelvic surgery, mesh procedures, inflammatory pelvic conditions or sensitisation within the sacral nerve pathways.
Key Clinical Message: Pudendal neuralgia describes pain involving the pudendal nerve. It does not automatically prove true nerve entrapment. In many patients, nerve irritation and sensitisation are more clinically relevant than fixed compression.
Pudendal Neuralgia and the Nantes Criteria
The classic Nantes criteria describe pudendal neuralgia caused by pudendal nerve entrapment. The typical pattern includes pain in the anatomical territory of the pudendal nerve, worsening with sitting, relief when standing or lying down, no objective sensory loss on examination, and pain relief after a diagnostic pudendal nerve block.
In practice, many patients have pudendal nerve involvement without fulfilling a strict entrapment pattern. They may describe burning, tingling, rawness, electric pain, rectal or vaginal foreign-body sensation, clitoral or vulval pain, perineal pain, pain after intercourse, pain after cycling, or sitting intolerance. These symptoms may coexist with pelvic floor myofascial pain, bladder pain syndrome, endometriosis, coccygeal pain or central sensitisation.
Pudendal Nerve Blocks: Diagnostic and Therapeutic Role
A pudendal nerve block can be useful when the clinical picture suggests pudendal nerve contribution. A positive diagnostic block, usually defined as meaningful temporary pain reduction, supports that the pudendal nerve is involved in the pain pathway. However, it does not by itself prove fixed entrapment, because local anaesthetic may reduce pain from irritation, inflammation, peripheral sensitisation or nearby muscular contributors.
Therapeutic pudendal nerve blocks may include local anaesthetic with or without steroid, depending on the clinical situation. Image guidance is important because the pudendal nerve is small, anatomically variable and closely related to vascular and pelvic structures. Blocks may be performed near the ischial spine, between the sacrospinous and sacrotuberous ligaments, or more distally in Alcock’s canal, depending on the suspected site of irritation.
When pudendal nerve pain persists despite physiotherapy, medication and diagnostic or therapeutic blocks, pulsed radiofrequency may be considered in carefully selected patients. Pulsed radiofrequency aims to modulate nerve signalling rather than destroy the nerve, making it more appropriate for non-malignant neuropathic pelvic pain than destructive neurolytic approaches.
Peripheral Nerve Irritation Syndromes: Clinical Clues
| Nerve / Region | Typical Pain Pattern | Assessment / Treatment Direction |
|---|---|---|
| Pudendal nerve | Perineal, vulval, vaginal, clitoral, rectal or anal pain; often worse with sitting and associated with sexual, urinary or bowel symptoms. | Pelvic floor assessment, Nantes criteria where relevant, image-guided pudendal nerve block, pulsed radiofrequency in selected refractory cases. |
| Ilioinguinal nerve | Lower abdominal, groin, mons pubis or labial pain, often after abdominal, hernia, gynaecological or caesarean surgery. | Examine scars and abdominal wall; consider ultrasound-guided diagnostic block or hydrodissection when clinically appropriate. |
| Iliohypogastric nerve | Suprapubic, lower abdominal or groin pain, sometimes overlapping with abdominal wall trigger points. | Carnett test, scar assessment, ultrasound-guided nerve block if the pain follows the expected distribution. |
| Genitofemoral nerve | Burning or radiating pain into the groin, mons pubis, labia majora or upper medial thigh; may overlap with iliopsoas dysfunction. | Assess iliopsoas, lumbar plexus irritation and prior surgery; consider targeted diagnostic block. |
| Posterior femoral cutaneous nerve | Posterior thigh, buttock, perineal or sitting-related pain; may mimic pudendal neuralgia or deep gluteal syndrome. | Assess sitting intolerance, deep gluteal tenderness and sciatic-type symptoms; consider image-guided diagnostic block. |
| Cluneal nerves | Low back, iliac crest, buttock or posterior pelvic pain that may be mistaken for sacroiliac joint or lumbar spine pain. | Palpation over the iliac crest and posterior pelvis; diagnostic nerve block when pain is focal and reproducible. |
Other peripheral neuralgias should be considered when pain follows a recognisable nerve distribution, has burning or electric qualities, is associated with numbness, tingling, allodynia or hypersensitivity, or began after surgery, trauma, cycling, childbirth or prolonged compression. These neuralgias often coexist with myofascial dysfunction rather than replacing it as a diagnosis.
The key treatment principle is to identify the dominant pain mechanism. A nerve block may clarify whether a nerve is contributing to pain, but durable improvement usually requires treating the surrounding muscle dysfunction, pelvic floor hypertonicity, visceral drivers and central sensitisation at the same time.
Visceral Pain and Visceral Sensitisation
Not all chronic pelvic pain arises from muscles or peripheral nerves. In many women, the original driver is persistent pain originating from pelvic organs such as the uterus, ovaries, bladder, bowel or surrounding peritoneum. Even when the underlying disease has been treated, the nervous system may remain sensitised, allowing organ-related pain to continue long after visible pathology has improved.
This process is known as visceral sensitisation. Repeated nociceptive input from endometriosis, adenomyosis, bladder pain syndrome, irritable bowel syndrome, pelvic inflammatory disease or postoperative adhesions can lower activation thresholds within peripheral nerves, the spinal cord and higher pain-processing centres. As a result, normally non-painful sensations such as bladder filling, bowel distension, intercourse or menstrual cramping may become disproportionately painful.
Key Clinical Message: Persistent pelvic pain does not always indicate ongoing tissue damage. In many women, the pelvic organs and nervous system have become sensitised, causing ordinary physiological sensations to be interpreted as pain.
Autonomic Contributions to Pelvic Pain
The pelvic organs are richly innervated by sympathetic and parasympathetic pathways. When these autonomic circuits become dysregulated, patients may experience urinary urgency, bladder pressure, bowel cramping, altered motility, sweating, nausea and heightened pain responses. Sympathetic overactivity can amplify nociceptive signalling and contribute to the sense of pelvic tension, pressure and persistent visceral discomfort.
This autonomic component helps explain why stress, sleep disturbance and emotional arousal often exacerbate pelvic pain. It also provides the rationale for interventional procedures such as superior hypogastric plexus block, inferior hypogastric plexus block and ganglion impar block, which aim to modulate abnormal sympathetic input from the pelvis.
Cross-Organ Sensitisation
Pelvic organs share overlapping sensory pathways within the spinal cord. Persistent nociceptive input from one organ can therefore increase sensitivity in another, a phenomenon known as cross-organ sensitisation. For example, endometriosis may heighten bladder and bowel sensitivity, while bladder pain syndrome may increase uterine or rectal pain.
This mechanism explains why many women have combinations of dysmenorrhoea, urinary urgency, frequency, painful bladder filling, constipation, bloating, dyspareunia and rectal pain. The coexistence of multiple diagnoses does not necessarily indicate separate diseases; it may reflect a shared sensitised pelvic pain network.
Central Amplification
When visceral pain persists over months or years, repeated nociceptive input can alter central nervous system processing. Pain pathways become more responsive, inhibitory mechanisms become less effective and the brain assigns greater salience to pelvic sensations. This process, often referred to as central amplification or central sensitisation, can lead to widespread pain, fatigue, sleep disturbance, cognitive symptoms and increased sensitivity to touch, pressure and movement.
Central amplification helps explain why some patients continue to experience significant pain despite apparently successful treatment of endometriosis or other pelvic pathology. It also explains why treatment often requires a multidimensional approach addressing the pelvic organs, muscles, nerves, autonomic system and central pain processing.
How Visceral Sensitisation Presents in Clinical Practice
| Mechanism | Clinical Features | Treatment Implications |
|---|---|---|
| Visceral sensitisation | Pain with bladder filling, bowel distension, menstruation, intercourse or organ movement. | Treat residual organ pathology, reduce inflammation and address nociceptive drivers. |
| Autonomic dysregulation | Urgency, cramping, sweating, nausea, stress-related flares and heightened physiological arousal. | Breathing strategies, nervous system regulation and sympathetic plexus interventions in selected cases. |
| Cross-organ sensitisation | Overlap of bladder, bowel, uterine and rectal symptoms. | Integrated multidisciplinary treatment rather than single-organ therapy. |
| Central amplification | Widespread pain, sleep disturbance, fatigue and heightened sensitivity. | Pain education, graded rehabilitation, medication and nervous system-focused therapies. |
The practical implication is that chronic pelvic pain often reflects a sensitised pelvic network rather than a single organ disorder. Successful treatment usually requires addressing ongoing visceral drivers while simultaneously treating autonomic dysregulation, myofascial dysfunction, peripheral nerve irritation and central amplification.
Interventional Treatments for Chronic Pelvic Pain Syndrome: A Stepwise Treatment Algorithm
Interventional treatment for chronic pelvic pain syndrome should be guided by the dominant pain mechanism rather than by symptoms alone. In many women, several mechanisms coexist: myofascial pain, pelvic floor hypertonicity, peripheral nerve irritation, visceral sensitisation, sacroiliac or coccygeal pain, and central amplification. The purpose of interventional treatment is therefore not simply to “block pain”, but to identify the relevant pain generators, reduce peripheral input, facilitate rehabilitation and interrupt the cycle of sensitisation.
The source article emphasises a stepwise approach. Pelvic floor physiotherapy and rehabilitation remain the foundation for myofascial CPPS. Image-guided injections and nerve blocks are most useful when they are targeted to examination findings and used to support active rehabilitation, rather than being used as isolated procedures.
Key Clinical Message: Interventions work best when they are used as part of an “injection-facilitated rehabilitation” model: identify the pain generator, reduce pain enough to allow movement and pelvic floor down-training, then reinforce the improvement with physiotherapy and nervous system regulation.
Stepwise Interventional Treatment Pathway
| Step | Treatment Focus | Clinical Use |
|---|---|---|
| Step 1 | Pelvic floor physiotherapy and rehabilitation | First-line foundation for myofascial CPPS, including internal and external myofascial release, down-training, biofeedback and neuromuscular re-education. |
| Step 2 | Trigger point injections | Used when specific pelvic floor, abdominal wall, hip or deep gluteal trigger points reproduce the patient’s familiar pain, especially when progress with physiotherapy is limited by pain. |
| Step 3 | Botulinum toxin for persistent hypertonicity | Considered for refractory pelvic floor hypertonicity, recurrent trigger points, obturator internus spasm or selected piriformis-related pain where repeated local anaesthetic injections give only short-lived benefit. |
| Step 4 | Peripheral nerve blocks | Used diagnostically and therapeutically when pain follows a neuropathic pattern, such as pudendal, ilioinguinal, iliohypogastric, genitofemoral, posterior femoral cutaneous or cluneal nerve irritation. |
| Step 5 | Pulsed radiofrequency | Considered when diagnostic blocks confirm a relevant nerve pathway but pain relief is short-lived; commonly considered for pudendal nerve, ganglion impar, selected peripheral nerves or dorsal root ganglion targets. |
| Step 6 | Ganglion impar block | Used for coccygeal, perineal, distal rectal, vulval, distal vaginal or anal pain, particularly where symptoms suggest sympathetic or deep visceral-perineal contribution. |
| Step 7 | Superior or inferior hypogastric plexus blocks | Reserved for selected patients with a strong visceral pelvic pain component, particularly midline pelvic pain, dysmenorrhoea-type pain, endometriosis-related visceral pain or refractory lower pelvic visceral pain. |
| Step 8 | Advanced neuromodulation or lesioning options | Includes cooled radiofrequency for selected sacroiliac-mediated pain and cryoneurolysis for selected peripheral nerve pain, especially where thermal radiofrequency is unsuitable. |
Trigger Point Injections
Trigger point injections are one of the most practical interventional treatments for myofascial CPPS. They may be used for pelvic floor muscles, abdominal wall trigger points, obturator internus, piriformis, iliopsoas, adductors and other muscles identified during examination. The source article highlights that benefit can occur regardless of the injectate used, suggesting that accurate needling of the trigger point itself may be therapeutic.
Local anaesthetic alone is often sufficient for diagnostic and therapeutic trigger point work. Steroid may be considered selectively, but it is not always necessary. Ultrasound guidance is particularly useful for deeper targets such as iliopsoas, piriformis and externally approached obturator internus, where adjacent nerves and vessels must be avoided.
Botulinum Toxin for Pelvic Floor Hypertonicity
Botulinum toxin may be considered when pelvic floor hypertonicity is persistent, function-limiting and refractory to physiotherapy and local anaesthetic trigger point injections. It is most relevant when the dominant problem is sustained muscle overactivity rather than isolated tenderness. The aim is to reduce excessive resting activity and create a window in which pelvic floor rehabilitation can progress.
The article notes that evidence is mixed, and botulinum toxin should therefore be reserved for carefully selected patients rather than used routinely. It is particularly considered when patients require repeated trigger point injections, have no durable improvement with local anaesthetic injections, or have practical barriers to frequent clinic visits and physiotherapy.
Peripheral Nerve Blocks
Peripheral nerve blocks have both diagnostic and therapeutic roles. They are most useful when symptoms suggest a recognisable nerve contribution, such as pudendal, ilioinguinal, iliohypogastric, genitofemoral, posterior femoral cutaneous or cluneal nerve irritation. A positive block supports nerve involvement, but it does not necessarily prove fixed entrapment.
For example, a pudendal nerve block may help confirm that the pudendal nerve is contributing to perineal, vulval, vaginal, clitoral, rectal or sitting-related pain. However, the block response must be interpreted alongside pelvic floor tone, obturator internus tenderness, coccygeal pain, surgical history and central sensitisation. Durable benefit usually requires treatment of the surrounding myofascial and visceral contributors as well as the nerve.
Pulsed Radiofrequency
Pulsed radiofrequency is a neuromodulation technique used to alter pain signalling without intentionally destroying the nerve. It may be considered when a diagnostic block identifies a relevant nerve pathway but the benefit from local anaesthetic is temporary. In CPPS, potential targets include the pudendal nerve, ganglion impar, selected peripheral nerves and, in specialist practice, dorsal root ganglion targets.
This approach is especially relevant in non-malignant pelvic pain because destructive neurolytic procedures are usually avoided unless there is a clear indication. Pulsed radiofrequency may be considered as part of a wider plan that includes pelvic floor therapy, medication optimisation and treatment of sensitisation.
Ganglion Impar Block
The ganglion impar is an important target for coccygeal, perineal, distal rectal, anal, vulval, distal urethral and distal vaginal pain. It lies anterior to the sacrococcygeal junction and forms the terminal part of the sympathetic chain. In the article, ganglion impar block is discussed as a particularly relevant intervention for coccydynia and chronic perineal pain.
A diagnostic ganglion impar block may help confirm whether this pathway is contributing to pain. For persistent non-malignant coccygeal or perineal pain, pulsed radiofrequency of the ganglion impar may be considered where repeated blocks give only short-lived benefit. Neurolytic procedures are generally more relevant to malignant pain and should be approached cautiously in benign chronic pain conditions.
Interventional Treatment Summary
| Procedure | Best Fit | Role in Treatment |
|---|---|---|
| Trigger point injections | Reproducible myofascial pain in pelvic floor, abdominal wall, obturator internus, piriformis, iliopsoas or adductors. | Diagnostic and therapeutic; best combined with immediate or ongoing physiotherapy. |
| Botulinum toxin | Persistent high-tone pelvic floor dysfunction or refractory muscle spasm. | Reduces excessive muscle activity; creates a rehabilitation window in selected patients. |
| Peripheral nerve blocks | Pudendal, ilioinguinal, iliohypogastric, genitofemoral, posterior femoral cutaneous or cluneal nerve irritation. | Clarifies nerve contribution and may reduce peripheral sensitisation. |
| Pulsed radiofrequency | Confirmed nerve-mediated pain with short-lived benefit from diagnostic blocks. | Neuromodulatory option for selected peripheral nerve, pudendal, DRG or ganglion impar targets. |
| Ganglion impar block | Coccydynia, perineal pain, distal rectal, anal, vulval or distal vaginal pain. | Diagnostic and therapeutic sympathetic/perineal pain intervention; pulsed RF may be considered if benefit is short-lived. |
| Superior hypogastric plexus block | Diffuse midline visceral pelvic pain, endometriosis-related visceral pain, adenomyosis, dysmenorrhoea-type pain or pelvic malignancy pain. | Specialist diagnostic and therapeutic block for confirmed visceral pelvic pain. |
| Inferior hypogastric plexus block | Lower pelvic visceral pain involving bladder base, lower rectum or vagina, especially if refractory to superior hypogastric plexus block. | Less commonly performed specialist intervention because of technical complexity. |
| Cooled radiofrequency | Selected sacroiliac joint-mediated posterior pelvic pain after positive diagnostic response. | Advanced option that creates a larger lesion zone for sacral lateral branch targets. |
| Cryoneurolysis | Selected peripheral nerve pain where a reversible, non-thermal technique is preferred. | May be useful when radiofrequency is unsuitable, including selected patients with implanted devices where thermal RF is avoided. |
Superior and Inferior Hypogastric Plexus Blocks
Superior and inferior hypogastric plexus blocks are specialist interventions for visceral pelvic pain. The superior hypogastric plexus lies anterior to L5-S1 near the aortic bifurcation and carries pain input from structures including the uterus, cervix, proximal fallopian tubes, bladder dome, sigmoid colon and rectum. It is most relevant for midline visceral pelvic pain, including selected cases of endometriosis-related pain, adenomyosis, refractory dysmenorrhoea-type pain and pelvic malignancy pain.
The inferior hypogastric plexus is located deeper in the presacral region around S2-S4 and is less commonly targeted because access is technically more difficult. It may be considered when lower pelvic visceral pain involves the bladder base, lower rectum or vagina, particularly when superior hypogastric plexus block does not adequately cover the pain pattern.
Important Selection Principle: Sympathetic plexus blocks should be considered only when there is a convincing visceral pain component. Pain that is focal, reproducible with muscle palpation, strongly positive on Carnett testing, or clearly dermatomal is less likely to respond to hypogastric plexus blockade.
Cooled Radiofrequency and Cryoneurolysis
Cooled radiofrequency is not a first-line pelvic pain treatment, but it may have a role in carefully selected posterior pelvic pain, particularly sacroiliac joint-mediated pain confirmed by diagnostic blocks. Its advantage is the creation of a larger treatment zone, which may be useful for sacral lateral branch targets where nerve anatomy can vary.
Cryoneurolysis is another advanced option for selected peripheral nerve pain. It is non-thermal and may be considered where conventional radiofrequency is unsuitable or where a reversible neuromodulatory approach is preferred. It should be reserved for clearly identified peripheral nerve pain rather than non-specific pelvic pain.
The overall principle is careful matching of treatment to pain mechanism. Trigger point injections and botulinum toxin target myofascial pain and hypertonicity; peripheral nerve blocks and pulsed radiofrequency target nerve irritation; ganglion impar and hypogastric plexus blocks target sympathetic or visceral pain; cooled radiofrequency and cryoneurolysis are reserved for selected refractory pain patterns where diagnostic blocks support the target.
Botulinum Toxin for Refractory Pelvic Floor Hypertonicity and Myofascial Pelvic Pain
Botulinum toxin is one of the most valuable advanced treatments for carefully selected women with chronic pelvic pain syndrome, particularly when persistent pelvic floor hypertonicity remains a major driver of symptoms. At Pain Spa, this is a key area of expertise and is used as part of a broader strategy to reduce muscle overactivity, facilitate pelvic floor rehabilitation and interrupt the cycle of pain sensitisation.
This treatment is not simply a “painkiller”. Botulinum toxin temporarily reduces excessive muscle contraction by blocking acetylcholine release at the neuromuscular junction. In the context of CPPS, the goal is to relax muscles that remain chronically overactive, reduce trigger point activity and create a therapeutic window during which pelvic floor physiotherapy becomes more effective.
Key Clinical Message: Botulinum toxin does not “cure” chronic pelvic pain. Its main role is to reduce persistent muscle overactivity so that the nervous system can calm down and pelvic floor rehabilitation can progress.
Who May Benefit?
Botulinum toxin is most useful when the dominant pain mechanism is persistent myofascial hypertonicity rather than active inflammation or untreated visceral disease. Typical candidates include women with:
✔ High-tone pelvic floor dysfunction with marked resting tension
✔ Recurrent trigger points despite physiotherapy
✔ Obturator internus spasm and pelvic sidewall pain
✔ Piriformis-related buttock and pelvic pain
✔ Severe dyspareunia related to pelvic floor overactivity
✔ Persistent urinary urgency, rectal pressure or outlet-type constipation driven by pelvic floor spasm
✔ Short-lived benefit from local anaesthetic trigger point injections
Target Muscles
Depending on examination findings, botulinum toxin may be directed to the levator ani complex (pubococcygeus, puborectalis and iliococcygeus), coccygeus, obturator internus, piriformis and other deep muscles that reproduce the patient’s familiar pain. The exact pattern of treatment should be individualised rather than applied according to a standard template.
Why Image Guidance Matters
The source article highlights the importance of accurate targeting. Deep muscles such as the obturator internus, piriformis and iliopsoas lie close to important nerves and blood vessels. Ultrasound guidance allows real-time visualisation of the needle tip and improves confidence that the treatment is delivered to the intended muscle rather than adjacent structures.
This precision is particularly important when symptoms involve a combination of pelvic floor dysfunction, pudendal nerve irritation and deep gluteal pain, because treatment success depends on targeting the true pain generator rather than relying on anatomical assumptions alone.
Expected Outcomes
When patient selection is appropriate, botulinum toxin may reduce pelvic floor tension, decrease trigger point activity, improve tolerance of intercourse, lessen urinary urgency and rectal pressure, and allow pelvic floor physiotherapy to progress more effectively. Benefits generally emerge gradually over several days to weeks.
As emphasised in your article, published evidence is mixed. Some patients experience substantial and prolonged improvement, while others derive limited benefit. For this reason, botulinum toxin is best reserved for carefully selected refractory cases rather than used routinely as first-line therapy.
Typical Advantages and Limitations
| Potential Advantages | Important Limitations |
|---|---|
| Reduces persistent muscle overactivity and trigger point activity. | Does not address untreated visceral pathology or central sensitisation on its own. |
| Creates a window for more effective pelvic floor physiotherapy. | Clinical response is variable and not all patients benefit. |
| May improve dyspareunia, urinary symptoms and pelvic pressure. | Effects are temporary and treatment may need repeating. |
| Useful when repeated local anaesthetic injections provide only short-lived benefit. | Should be performed by clinicians experienced in pelvic anatomy and image-guided techniques. |
Pain Spa Expert Perspective
At Pain Spa, Dr Krishna uses advanced ultrasound-guided techniques to target deep pelvic and gluteal muscles with high precision. Careful assessment is used to determine whether persistent pelvic floor hypertonicity is the dominant driver of symptoms and whether botulinum toxin is likely to add meaningful benefit beyond physiotherapy and standard injections.
When used in the right patient and integrated into a structured rehabilitation programme, botulinum toxin can be a highly valuable tool for women with severe refractory pelvic floor dysfunction and chronic myofascial pelvic pain.
Low-Dose Naltrexone (LDN) for Nociplastic Pain and Neuroinflammation
Low-dose naltrexone (LDN) is an emerging treatment option for patients with chronic pelvic pain syndrome in whom central sensitisation, nociplastic pain and neuroinflammation appear to play a significant role. Unlike conventional analgesics, LDN is not used to suppress pain directly. Instead, it is thought to modulate immune and nervous system activity, helping to reduce the persistent amplification of pain signals.
In chronic pelvic pain, pain may persist even after identifiable structural problems such as endometriosis, adhesions or pelvic floor trigger points have been treated. In this situation, ongoing activation of glial cells and inflammatory signalling within the central nervous system may contribute to widespread sensitivity, fatigue, sleep disturbance, brain fog and heightened responses to normal bodily sensations. The article highlights LDN as a useful option when these nociplastic mechanisms are prominent.
Key Clinical Message: Low-dose naltrexone is not a conventional painkiller. Its main role is to reduce abnormal nervous system sensitisation and neuroinflammatory signalling in patients with chronic pain that has become “stuck on.”
How Low-Dose Naltrexone (LDN) Works
At the very low doses used in chronic pain, naltrexone appears to have effects that are distinct from its standard use in addiction medicine. Proposed mechanisms include modulation of microglial activation, reduction in pro-inflammatory cytokine signalling and restoration of more balanced endogenous opioid activity. The overall effect may be a gradual reduction in central amplification and improved regulation of the nervous system.
Because these mechanisms target pain processing rather than structural pathology, LDN may be particularly useful in women with overlapping conditions such as fibromyalgia, irritable bowel syndrome, chronic fatigue, migraine, widespread pain, post-viral syndromes and other disorders characterised by nervous system hypersensitivity.
When LDN May Be Helpful
LDN may be considered when:
✔ Pelvic pain persists despite appropriate treatment of myofascial, neural and visceral contributors.
✔ Symptoms suggest prominent central sensitisation or nociplastic pain.
✔ The patient also has fibromyalgia, chronic fatigue, migraine, irritable bowel syndrome or widespread pain.
✔ Conventional medications have been poorly tolerated or ineffective.
✔ A more nervous-system-focused treatment approach is desired.
Dosing and Practical Considerations
The article describes LDN as a treatment that is usually introduced gradually. Many clinicians begin with a very low dose, such as 0.5 mg to 1.5 mg taken at night, and increase slowly over several weeks depending on tolerability and clinical response. Common maintenance doses range between 3 mg and 4.5 mg daily, although individual requirements vary.
Improvement is typically gradual rather than immediate. Some patients notice benefits within a few weeks, whereas others require two to three months before a meaningful change becomes apparent. Temporary vivid dreams, sleep disturbance, headache or gastrointestinal symptoms may occur, but these effects are usually mild and can often be managed by slower dose escalation or adjusting the timing of administration.
Because naltrexone blocks opioid receptors, LDN is generally not suitable for patients who require regular opioid medication. This issue should be discussed carefully before treatment is started.
Potential Advantages and Limitations
| Potential Advantages | Important Limitations |
|---|---|
| Targets central sensitisation and neuroinflammatory mechanisms. | Clinical evidence is still evolving. |
| Usually well tolerated at low doses. | Benefits may take several weeks or months to emerge. |
| May help patients with overlapping nociplastic conditions. | Generally unsuitable for patients taking regular opioid medication. |
| Can be combined with physiotherapy and interventional treatments. | Not a substitute for treating myofascial, neural or visceral pain generators. |
Available Through Pain Spa
Low-dose naltrexone is available through Pain Spa following a comprehensive assessment by Dr Krishna. Treatment is prescribed only when the clinical picture suggests that nociplastic pain, central sensitisation and neuroinflammation are important contributors to the patient’s symptoms.
For carefully selected patients, low-dose naltrexone can be a valuable part of a broader treatment plan that also addresses myofascial dysfunction, peripheral nerve irritation, visceral sensitisation, sleep disturbance and nervous system dysregulation.
Medication-Based Treatments for Chronic Pelvic Pain Syndrome
Medication-based treatment in chronic pelvic pain syndrome should be guided by the dominant pain mechanism. Some medications aim to reduce neuropathic pain and central sensitisation, while others reduce hormonally driven nociceptive input from conditions such as endometriosis and adenomyosis. Medication is rarely sufficient on its own, but it can be an important part of a wider plan that includes pelvic floor therapy, psychological support and targeted interventions.
Key Clinical Message: Medications should be matched to the pain mechanism: neuromodulators for nerve irritation and central amplification, hormonal treatment for cyclic or endometriosis-related pain, and topical treatments for localised vulval, scar or neuropathic pain.
Neuropathic Pain Medications
Neuropathic pain medications may be considered when symptoms suggest nerve irritation, burning pain, electric shocks, tingling, allodynia, hypersensitivity, widespread pain or central sensitisation. These medicines do not “numb” the pelvis. Their role is to reduce abnormal pain signalling and improve tolerance of movement, touch, sitting, pelvic floor therapy and rehabilitation.
Neuropathic Pain Medication Options
| Medication Group | Potential Role | Important Considerations |
|---|---|---|
| SNRIs Duloxetine, venlafaxine |
May help neuropathic pain, widespread pain, mood symptoms and central sensitisation. | Evidence is extrapolated from other neuropathic pain conditions; no strong CPPS-specific trial evidence for duloxetine or venlafaxine. |
| Tricyclic antidepressants Amitriptyline, nortriptyline, desipramine |
May help sleep, neuropathic pain, pain amplification and coexisting mood symptoms. | Start low and increase slowly; sedation, dry mouth, constipation and cognitive effects may limit tolerability. |
| Gabapentinoids Gabapentin, pregabalin |
May be useful for selected neuropathic pain presentations. | The GaPP2 trial showed gabapentin was not superior to placebo for chronic pelvic pain without obvious pelvic pathology and was associated with more adverse effects. Use should be selective rather than routine. |
| Topical treatments Lidocaine or compounded topical agents |
May help localised vulval, scar-related or superficial neuropathic pain. | Best suited to localised symptoms; less useful for deep pelvic, visceral or widespread sensitised pain. |
The article emphasises that neuropathic medications should not replace assessment for myofascial dysfunction. Nerve pain and muscle pain often coexist. Reducing sensory hypersensitivity may improve the patient’s ability to tolerate pelvic floor physiotherapy, but untreated trigger points, pelvic floor hypertonicity or visceral drivers still need to be addressed directly.
Hormonal Therapies
Hormonal therapies are most relevant when pain has a cyclic pattern or when endometriosis, adenomyosis or menstrual exacerbation is suspected. The aim is to reduce hormonally driven nociceptive input from pelvic organs, particularly where dysmenorrhoea, deep dyspareunia, cyclic bowel symptoms or cyclic bladder symptoms are prominent.
Hormonal Treatment Options
| Treatment | Typical Role | Practical Considerations |
|---|---|---|
| Continuous combined oral contraceptives | First-line option for cyclic pelvic pain and menstrual exacerbation where appropriate. | Continuous use may be more useful than cyclic use for dysmenorrhoea suppression. |
| Progestogen-based therapy | Used for endometriosis-related pain, adenomyosis and menstrual suppression. | Choice depends on tolerability, bleeding profile, contraindications and gynaecology input. |
| GnRH agonists | Specialist option for endometriosis-related pain where first-line hormonal therapy is inadequate. | May require add-back therapy and monitoring because of hypo-oestrogenic adverse effects. |
| GnRH antagonists | Oral specialist treatment option for endometriosis-associated pain in selected patients. | Treatment duration, bone health, side effects and add-back therapy need careful consideration. |
| Endometriosis-directed treatment | Reduces ongoing visceral nociceptive input from active or suspected endometriosis. | Pain may persist if pelvic floor myalgia, nerve irritation or central sensitisation are not treated at the same time. |
The source article highlights an important limitation: treating the hormonal or gynaecological driver alone may not resolve pain once pelvic floor hypertonicity, nerve irritation and sensitisation have become established. A patient may have good suppression of endometriosis activity yet continue to experience pain because the pelvic muscles and nervous system remain sensitised.
Practical Treatment Principle: Hormonal therapy may reduce the visceral driver, while neuromodulators may reduce pain amplification. For many women, both need to be combined with pelvic floor treatment and carefully selected interventional procedures.
Why Opioids Are Not Recommended
The article is clear that opioids are not recommended for chronic pelvic pain syndrome. They do not address the underlying mechanisms of CPPS and carry significant risks including tolerance, dependence, opioid-induced hyperalgesia, sedation, constipation and reduced long-term function. Patients already taking opioids should usually be reviewed carefully and, where appropriate, supported through gradual reduction rather than abrupt cessation.
Medication decisions should therefore be individualised and mechanism-based. The best results are usually achieved when medicines are used to support rehabilitation, reduce sensitisation and treat active visceral drivers, rather than being used as the sole treatment for chronic pelvic pain.
Pelvic Floor Rehabilitation, Psychological Therapies and Trauma-Informed Care
Rehabilitation is central to the treatment of chronic pelvic pain syndrome. In many women, pain is maintained by a combination of pelvic floor guarding, myofascial trigger points, nervous system hypervigilance, fear of symptom flares, poor sleep, trauma-related responses and central sensitisation. For this reason, treatment should not focus only on injections or medication. Long-term improvement usually requires pelvic floor rehabilitation, nervous system regulation and trauma-sensitive care working together.
The aim is not simply to strengthen the pelvic floor. In high-tone pelvic floor dysfunction, the problem is often excessive resting tone, impaired relaxation and protective guarding. The priority is therefore usually down-training, myofascial release, improved relaxation, graded exposure to movement and restoration of confidence in normal pelvic function.
Key Clinical Message: Pelvic floor rehabilitation and psychological therapies are not used because the pain is “in the mind”. They are used because chronic pelvic pain changes muscle tone, threat perception, autonomic arousal and central pain processing.
Pelvic Floor Physical Therapy
Pelvic floor physical therapy is a first-line treatment for myofascial CPPS and high-tone pelvic floor dysfunction. Treatment may include internal and external myofascial release, trigger point therapy, breathing retraining, pelvic floor relaxation, hip and abdominal wall work, biofeedback and neuromuscular re-education. The focus is on restoring the patient’s ability to voluntarily relax the pelvic floor rather than repeatedly bracing or guarding.
Biofeedback can be helpful when patients cannot sense whether the pelvic floor is relaxed or contracted. Dilator therapy may be useful when dyspareunia, vaginismus-type guarding or fear of penetration is present, but it should be introduced gradually and only within a supportive, consent-based rehabilitation plan.
Core Components of Pelvic Floor Rehabilitation
| Treatment Component | Purpose |
|---|---|
| Down-training | Reduces excessive resting tone and teaches the pelvic floor to relax rather than guard. |
| Myofascial release | Targets trigger points, taut bands and restricted tissues in the pelvic floor, abdominal wall, hip and gluteal muscles. |
| Biofeedback | Helps patients recognise and reduce involuntary pelvic floor contraction. |
| Dilator therapy | Supports graded exposure where penetration pain, vaginismus-type guarding or fear-related tightening is present. |
| Graded movement | Restores confidence with walking, sitting, exercise, cycling or sexual activity without repeated flare cycles. |
Psychological Therapies and Nervous System Regulation
Psychological therapies are used in CPPS because pain, threat perception and autonomic arousal become linked over time. Persistent pain can lead to fear of movement, fear of intercourse, avoidance, sleep disturbance, low mood, anxiety, hypervigilance and reduced confidence. These responses are understandable, but they can also keep the nervous system in a protective, sensitised state.
Cognitive behavioural therapy can help patients identify unhelpful fear-avoidance patterns and build coping strategies. Acceptance and commitment therapy focuses on improving function and quality of life even when symptoms fluctuate. Mindfulness-based stress reduction may help reduce autonomic arousal and pain reactivity. Emotional Awareness and Expression Therapy may be particularly relevant where pain is associated with unresolved stress, emotional conflict or trauma-related nervous system activation.
Psychological Therapy Options in CPPS
| Therapy | Main Focus | Clinical Relevance |
|---|---|---|
| CBT | Pain coping, flare planning, behavioural activation and fear-avoidance patterns. | Useful when pain has led to avoidance, reduced activity, anxiety or loss of confidence. |
| ACT | Psychological flexibility, values-based activity and acceptance of fluctuating symptoms. | Useful when the goal is improved function despite ongoing pain sensitivity. |
| MBSR | Mindfulness, body awareness and reduction of stress-related arousal. | Useful when stress, sleep disturbance and autonomic arousal worsen flares. |
| EAET | Emotional processing, unresolved conflict and trauma-linked pain mechanisms. | May be particularly relevant in patients with nociplastic pain, trauma history, PTSD symptoms or complex emotional stressors. |
Trauma-Informed Care
Trauma-informed care is essential in chronic pelvic pain because pelvic examinations, internal physiotherapy, sexual pain discussions and previous medical experiences may be distressing for some patients. A trauma-informed approach does not assume that trauma is the cause of pain. It simply recognises that many people with chronic pain have experienced trauma, and that safety, control and trust can strongly influence treatment tolerance.
Practical trauma-informed care includes explaining each step before it happens, asking permission, allowing the patient to pause or stop the examination, avoiding rushed internal assessment, using clear and non-judgemental language, and validating the reality of the patient’s pain. This is particularly important in patients with PTSD, previous abuse, difficult childbirth, surgical trauma or repeated invalidating healthcare encounters.
Principles of Trauma-Informed Pelvic Pain Care
| Principle | How It Helps |
|---|---|
| Choice and control | Reduces threat response and helps the patient feel safe during assessment and treatment. |
| Clear explanation | Prevents surprise, uncertainty and unnecessary anxiety during examination or procedures. |
| Permission at each stage | Allows internal examination or physiotherapy to proceed only when the patient is ready. |
| Validation | Reinforces that the pain is real and not imagined, even when imaging or laparoscopy is normal. |
| Integrated support | Combines pelvic floor therapy, psychological treatment, medication and interventions without separating mind and body. |
The practical message is that rehabilitation in CPPS must be paced, collaborative and mechanism-based. Pelvic floor therapy treats muscle guarding; psychological therapies address pain-related fear, avoidance and central amplification; trauma-informed care ensures that treatment itself does not become another source of threat. Together, these approaches provide the foundation on which medication and interventional treatments can work more effectively.
Stepwise Treatment Algorithm for Chronic Pelvic Pain Syndrome
Chronic pelvic pain syndrome should be treated using a structured, mechanism-based pathway. The aim is to identify the dominant contributors — visceral pain, pelvic floor hypertonicity, myofascial trigger points, peripheral nerve irritation, autonomic dysfunction and central sensitisation — and then escalate treatment in a logical sequence.
The article emphasises that treatment should not be purely linear. Many patients require several approaches at the same time. For example, pelvic floor physiotherapy may need to be combined with trigger point injections, neuromodulator medication, trauma-informed care and treatment of endometriosis or bladder pain syndrome.
Key Clinical Message: The best outcomes usually come from matching treatment to pain mechanism, not simply escalating from one procedure to another.
Practical Escalation Pathway
Step 1 — Identify and Treat Visceral Drivers
Assess for endometriosis, adenomyosis, bladder pain syndrome, irritable bowel syndrome, pelvic congestion, infection, inflammatory disease and postsurgical causes. Treating these drivers reduces ongoing nociceptive input into the pelvic pain system.
Step 2 — Start Pelvic Floor Rehabilitation
For myofascial CPPS and high-tone pelvic floor dysfunction, pelvic floor physiotherapy is a core treatment. This includes down-training, internal and external myofascial release, breathing work, biofeedback and graded functional rehabilitation.
Step 3 — Add Trigger Point Injections When Pain Blocks Progress
If specific muscles reproduce the patient’s familiar pain and physiotherapy is limited by pain, targeted trigger point injections may be used. These may involve pelvic floor muscles, obturator internus, piriformis, abdominal wall, iliopsoas or adductors.
Step 4 — Consider Botulinum Toxin for Refractory Hypertonicity
When pelvic floor overactivity remains persistent despite physiotherapy and local anaesthetic injections, botulinum toxin may be considered in carefully selected patients. The aim is to reduce excessive resting tone and create a rehabilitation window.
Step 5 — Treat Peripheral Nerve Irritation
When pain has burning, tingling, electric, dermatomal or sitting-related features, targeted nerve blocks may help identify pudendal, ilioinguinal, iliohypogastric, genitofemoral, posterior femoral cutaneous or cluneal nerve involvement.
Step 6 — Use Neuromodulation for Confirmed Persistent Nerve Pain
If diagnostic blocks confirm a relevant nerve pathway but relief is short-lived, pulsed radiofrequency may be considered for selected targets such as the pudendal nerve, ganglion impar, peripheral nerves or dorsal root ganglion.
Step 7 — Address Central Sensitisation and Nociplastic Pain
When symptoms include widespread pain, fatigue, sleep disturbance, brain fog, hypersensitivity or multiple overlapping pain syndromes, treatment should include pain education, psychological therapies, sleep optimisation, neuromodulator medication or low-dose naltrexone where appropriate.
Step 8 — Consider Visceral and Sympathetic Blocks in Selected Cases
For deep visceral pelvic pain, especially midline pain, dysmenorrhoea-type pain, endometriosis-related visceral pain, malignancy-related pain or refractory perineal pain, superior hypogastric plexus block, inferior hypogastric plexus block or ganglion impar block may be considered.
Step 9 — Reserve Advanced Procedures for Clearly Confirmed Targets
Cooled radiofrequency, cryoneurolysis, dorsal root ganglion-level interventions and other advanced treatments should be reserved for carefully selected patients where diagnostic blocks and clinical assessment clearly support the target.
Treatment Pathway Summary
| Dominant Pain Mechanism | Treatment Direction |
|---|---|
| Visceral pain | Treat endometriosis, adenomyosis, bladder pain syndrome, IBS or pelvic congestion; consider hypogastric plexus block only in selected refractory visceral pain. |
| Pelvic floor hypertonicity | Pelvic floor down-training, myofascial release, trigger point injections and botulinum toxin in refractory cases. |
| Peripheral nerve irritation | Targeted diagnostic nerve blocks, medication support and pulsed radiofrequency if block response is positive but short-lived. |
| Coccygeal or perineal pain | Coccygeal assessment, pelvic floor therapy, ganglion impar block or pulsed radiofrequency in selected cases. |
| Sacroiliac-mediated posterior pelvic pain | Rehabilitation, diagnostic sacroiliac joint injection and radiofrequency options only when diagnostic response supports the target. |
| Central sensitisation / nociplastic pain | Pain education, psychological therapies, sleep optimisation, neuromodulator medication and low-dose naltrexone where appropriate. |
The practical message is that CPPS treatment should be targeted, staged and integrated. Procedures are most effective when they are used to reduce pain enough for rehabilitation to progress, while medication and psychological therapies help calm the sensitised nervous system. Advanced interventions should be reserved for well-defined pain generators rather than used as a substitute for comprehensive assessment.
Muscle-by-Muscle Injection Table for Chronic Pelvic Pain Syndrome
A muscle-by-muscle approach helps translate examination findings into targeted treatment. The aim is not to inject every tender area, but to identify the muscles that reproduce the patient’s familiar pain and contribute to the wider pelvic pain pattern.
The source article recommends a structured sequence: first perform an external musculoskeletal assessment, then a single-digit internal pelvic floor assessment where appropriate, and finally correlate the symptom pattern with the relevant muscle group before planning treatment.
Key Clinical Message: The most useful injection target is the structure that reproduces the patient’s familiar pain, not simply the most tender point on examination.
Stepwise Assessment Before Injection
| Step | Assessment Focus | Purpose |
|---|---|---|
| Step 1 | External musculoskeletal assessment: gait, posture, abdomen, lower back, sacroiliac joints, pubic symphysis and hip adductors. | Identifies abdominal wall, hip, groin, sacroiliac and posterior pelvic contributors before assuming the pain is purely pelvic floor-related. |
| Step 2 | Targeted clinical tests such as Carnett test, FABER test, resisted hip flexion, resisted adduction and neurosensory mapping. | Helps distinguish myofascial pain, joint pain and peripheral nerve irritation. |
| Step 3 | Internal single-digit pelvic floor assessment where clinically appropriate and consented. | Assesses tone, trigger points, tenderness, ability to contract and relax, and reproduction of familiar pain from levator ani, obturator internus and coccygeus. |
| Step 4 | Symptom-to-muscle correlation. | Guides whether treatment should focus on physiotherapy, trigger point injection, botulinum toxin, nerve block, radiofrequency or combined care. |
Anterior and Lateral Targets
| Target | Symptoms / Clinical Pattern | Examination Clues | Treatment Direction |
|---|---|---|---|
| Rectus abdominis | Lower abdominal pain, scar-related pain after caesarean section or laparoscopy, pain mimicking visceral pelvic pain. | Pain at the lateral rectus border; positive Carnett test; pain worsened by sit-ups or abdominal tensing. | Abdominal wall physiotherapy, trigger point injection, scar assessment, ACNES or cutaneous nerve block if neuropathic features are present. |
| Internal oblique / transversus abdominis | Lower quadrant pain, pain after inguinal or abdominal surgery, pain with twisting or trunk rotation. | Tenderness lateral to rectus; pain with resisted trunk rotation; possible ilioinguinal or iliohypogastric nerve irritation. | External trigger point injection; ultrasound-guided ilioinguinal or iliohypogastric nerve block if nerve irritation is suspected. |
| Iliopsoas / iliacus | Deep anterior pelvic pain, groin pain, low back pain, pain with hip flexion, stairs, prolonged sitting or walking. | Positive Thomas test, pain with resisted hip flexion, deep abdominal or iliacus tenderness. | Targeted physiotherapy, stretching, ultrasound-guided trigger point injection; consider genitofemoral or lateral femoral cutaneous nerve involvement where symptoms fit. |
| Hip adductors | Medial groin pain, pubic pain, pain with walking, exercise, resisted adduction or pelvic loading. | Tenderness over adductor origin or muscle belly; pain with resisted adduction or adductor squeeze test. | Adductor rehabilitation, pelvic floor–adductor coordination work, trigger point injection, pubic symphysis assessment if pain is midline. |
| Pubic symphysis / adductor-rectus complex | Anterior groin or pubic pain, pain with coughing, Valsalva, sit-ups or athletic loading. | Pubic tenderness, pain with combined resisted adduction and sit-up, possible osteitis pubis or athletic pubalgia pattern. | Core and adductor rehabilitation, imaging if instability or osteitis pubis suspected, targeted injection in selected cases. |
Pelvic Floor and Deep Pelvic Targets
| Target | Symptoms / Clinical Pattern | Examination Clues | Treatment Direction |
|---|---|---|---|
| Levator ani complex Pubococcygeus, puborectalis, iliococcygeus |
Deep pelvic pain, pelvic heaviness, dyspareunia, rectal pressure, urinary urgency, pain after intercourse or prolonged sitting. | Internal palpation reproduces familiar pain; high tone; trigger points; impaired relaxation after contraction. | Pelvic floor down-training, internal myofascial release, trigger point injection, botulinum toxin in refractory hypertonicity. |
| Obturator internus | Pelvic sidewall pain, deep vaginal or rectal pain, hip or buttock pain, dyspareunia, sitting pain, urinary or bowel symptoms. | Tenderness on anterolateral vaginal wall palpation; pain with resisted external rotation; positive FABER may coexist. | Pelvic floor physiotherapy, obturator internus release, ultrasound-guided or carefully targeted injection, botulinum toxin in selected refractory cases. |
| Coccygeus | Posterior pelvic pain, rectal pressure, coccyx-related pain, pain with defaecation or sitting. | Posterior vaginal wall tenderness near the coccyx; coccygeal mobility restriction or pain; associated pelvic floor coordination difficulty. | Pelvic floor therapy, coccygeal mobilisation, trigger point injection, ganglion impar block if refractory and pain pattern fits. |
| Bulbospongiosus / ischiocavernosus | Superficial dyspareunia, vulval or vestibular pain, pain at the vaginal entrance. | Tenderness at the introitus; superficial palpation reproduces pain; cotton-swab testing may identify vestibular sensitivity. | Superficial pelvic floor release, topical treatments, graded dilator therapy, carefully selected trigger point injection. |
Posterior and Deep Gluteal Targets
| Target | Symptoms / Clinical Pattern | Examination Clues | Treatment Direction |
|---|---|---|---|
| Piriformis | Buttock pain, sitting intolerance, sciatica-like pain, pelvic pain with possible pudendal or sacral nerve irritation. | Deep gluteal tenderness; pain with hip rotation testing; symptoms provoked by sitting or piriformis loading. | Physiotherapy, ultrasound-guided piriformis injection, botulinum toxin in selected refractory cases, sciatic or pudendal evaluation if symptoms suggest neural irritation. |
| Gemelli / deep rotator complex | Deep buttock pain, posterior pelvic pain, deep gluteal syndrome-type symptoms. | Pain reproduced by deep gluteal palpation and hip rotation; may overlap with obturator internus and sciatic irritation. | Deep gluteal rehabilitation, ultrasound-guided diagnostic injection in selected cases, nerve assessment if symptoms radiate. |
| Quadratus femoris | Deep posterior hip or buttock pain, sometimes worse with hip extension or rotation. | Posterior hip tenderness, deep gluteal pain pattern, possible ischiofemoral impingement features. | Hip-focused rehabilitation, imaging if impingement suspected, image-guided injection where appropriate. |
| Sacroiliac joint / sacral lateral branches | Posterior pelvic pain below L5, unilateral buttock pain, postpartum pelvic girdle pain, pain with sit-to-stand transitions. | Fortin finger test; cluster of positive SIJ provocation tests; diagnostic SIJ block response. | Physiotherapy, SIJ belt where appropriate, intra-articular or periarticular injection, sacral lateral branch radiofrequency or cooled RF in selected confirmed cases. |
| Coccyx / sacrococcygeal joint | Tailbone pain, pain with sitting on hard surfaces, pain rising from sitting, pain with defaecation. | Direct coccygeal tenderness; pain on coccygeal mobility testing; dynamic imaging if instability suspected. | Cushioning, pelvic floor therapy, coccygeal mobilisation, sacrococcygeal injection, ganglion impar block or pulsed RF in refractory cases. |
Practical Treatment Principle: Begin with pelvic floor physiotherapy and rehabilitation for all myofascial CPPS. Add trigger point injections when pain limits progress. Consider botulinum toxin for persistent hypertonicity, nerve blocks when neuropathic features are present, and radiofrequency or ganglion impar procedures only when diagnostic assessment clearly supports the target.
This table should be used as a clinical map rather than a checklist. In CPPS, multiple muscles are often involved simultaneously, and the most successful treatment plan is usually one that treats the full myofascial chain while also addressing visceral drivers, peripheral nerve irritation and central sensitisation.
Pain Spa Expert Perspective and Treatments Offered
Chronic pelvic pain syndrome requires careful assessment of multiple overlapping pain mechanisms, including pelvic floor hypertonicity, myofascial trigger points, peripheral nerve irritation, visceral sensitisation, sacroiliac and coccygeal dysfunction, and central sensitisation. At Pain Spa, Dr M. Krishna brings extensive experience in advanced ultrasound-guided and fluoroscopy-guided pain procedures, allowing treatment to be targeted precisely to the structures most likely to be driving each patient’s symptoms.
The Pain Spa approach is based on identifying the dominant pain generators, confirming clinically relevant targets where appropriate, and integrating interventional treatments with pelvic floor rehabilitation, medication optimisation and nervous system-focused care. The goal is not simply to provide an injection, but to develop a comprehensive and individualised treatment strategy.
Pain Spa Expert Perspective: Successful treatment of chronic pelvic pain depends on accurate diagnosis, careful patient selection and precise image-guided treatment. This is particularly important for deep pelvic muscles, pudendal and other peripheral nerves, ganglion impar, hypogastric plexus and sacroiliac targets.
Treatments Offered at Pain Spa
✔ Comprehensive chronic pelvic pain assessment
✔ Ultrasound-guided trigger point injections
✔ Pelvic floor, obturator internus and piriformis-focused injections
✔ Botulinum toxin injections for refractory pelvic floor hypertonicity
✔ Pudendal nerve blocks and pulsed radiofrequency
✔ Ilioinguinal, iliohypogastric and genitofemoral nerve blocks
✔ Posterior femoral cutaneous and cluneal nerve blocks
✔ Ganglion impar block and pulsed radiofrequency
✔ Sacroiliac joint injections and radiofrequency treatment
✔ Superior hypogastric plexus block
✔ Inferior hypogastric plexus block
✔ Cooled radiofrequency
✔ Cryoneurolysis
✔ Low-dose naltrexone for central sensitisation and nociplastic pain
Patients with persistent pelvic pain, failed previous treatments or complex overlapping symptoms may benefit from specialist assessment at Pain Spa to determine whether image-guided interventional pain treatment could form part of a broader rehabilitation and recovery plan.
Frequently Asked Questions
Can chronic pelvic pain continue even after endometriosis has been treated?
Yes. Pain can persist if pelvic floor hypertonicity, myofascial trigger points, nerve irritation or central sensitisation have become established. This does not mean the pain is imaginary; it means the pain system has become sensitised and needs targeted treatment.
Is pelvic floor physiotherapy always about strengthening?
No. In high-tone pelvic floor dysfunction, the problem is often excessive resting tension rather than weakness. Treatment usually focuses on down-training, relaxation, myofascial release, breathing, biofeedback and graded return to normal activity.
Does pudendal neuralgia always mean pudendal nerve entrapment?
No. Pudendal neuralgia means pain involving the pudendal nerve. In many patients the nerve is irritated or sensitised rather than truly trapped. Pelvic floor spasm, obturator internus tightness, sitting pressure, surgery, cycling, childbirth and central sensitisation may all contribute.
What is the role of trigger point injections?
Trigger point injections can help when specific muscles reproduce the patient’s familiar pain. They are most useful when combined with pelvic floor physiotherapy, because the injection may reduce pain enough to allow better movement, relaxation and rehabilitation.
When is botulinum toxin considered?
Botulinum toxin may be considered in carefully selected patients with refractory pelvic floor hypertonicity, persistent muscle spasm or recurrent trigger points despite physiotherapy and local anaesthetic injections. It is used to reduce excessive muscle overactivity and create a window for rehabilitation.
Can low-dose naltrexone help chronic pelvic pain?
Low-dose naltrexone may help selected patients where nociplastic pain, central sensitisation and neuroinflammation appear to be important contributors. It is not a conventional painkiller and usually works gradually over weeks to months.
Are hypogastric plexus blocks suitable for all pelvic pain?
No. Superior or inferior hypogastric plexus blocks are most relevant when there is a strong visceral pelvic pain component, such as deep midline pelvic pain, dysmenorrhoea-type pain or selected endometriosis-related visceral pain. They are less likely to help pain that is mainly muscular, dermatomal or abdominal wall-related.
Why is treatment often multidisciplinary?
CPPS usually involves overlapping visceral, myofascial, neural and central pain mechanisms. A single treatment rarely addresses all contributors. Better outcomes usually come from combining pelvic floor rehabilitation, medication, psychological support and carefully selected image-guided interventions.
Key Clinical Take-Home Messages
Chronic pelvic pain syndrome is rarely caused by a single diagnosis or a single structure. In most women, symptoms arise from a combination of visceral pain, pelvic floor hypertonicity, myofascial trigger points, peripheral nerve irritation, autonomic dysregulation and central sensitisation. The most successful treatment plans are those that identify the dominant contributors and address them in a coordinated, mechanism-based way.
Essential Clinical Messages
✔ Chronic pelvic pain often persists because the nervous system, muscles and pelvic organs have become sensitised, even when imaging or laparoscopy is normal.
✔ Pelvic floor hypertonicity and myofascial trigger points are among the most common and most frequently overlooked contributors to persistent pelvic pain.
✔ Pudendal neuralgia usually reflects pudendal nerve irritation or sensitisation rather than fixed entrapment.
✔ Endometriosis and other visceral disorders can initiate pain, but treatment of the original pathology alone may not resolve established myofascial and central sensitisation.
✔ Pelvic floor physiotherapy focused on down-training and myofascial release is a cornerstone of treatment.
✔ Trigger point injections, botulinum toxin, nerve blocks and radiofrequency procedures are most effective when used to facilitate rehabilitation rather than as isolated treatments.
✔ Low-dose naltrexone and neuromodulator medications may help selected patients with prominent nociplastic pain and central sensitisation.
✔ Psychological therapies and trauma-informed care address fear, hypervigilance and autonomic arousal without implying that the pain is psychological.
✔ Opioids are generally not recommended because they do not address the underlying mechanisms of chronic pelvic pain and may worsen long-term outcomes.
✔ The best outcomes usually come from a multidisciplinary, mechanism-based approach tailored to the individual patient.
✔ Accurate diagnosis, careful patient selection and precise image-guided interventions can significantly improve outcomes in carefully selected patients.
The central message of this article is that chronic pelvic pain syndrome is understandable and treatable. Even when symptoms have persisted for years and multiple previous treatments have failed, meaningful improvement is often possible when treatment is directed at the correct pain mechanisms and integrated into a structured rehabilitation programme.
References
The following key guidelines, systematic reviews and clinical studies informed the content of this article and reflect the current understanding of chronic pelvic pain syndrome, pelvic floor dysfunction, peripheral nerve irritation, central sensitisation and interventional pain management.
1. American College of Obstetricians and Gynecologists (ACOG). Chronic Pelvic Pain. Practice Bulletin. Obstetrics & Gynecology. 2020.
2. European Association of Urology (EAU). Guidelines on Chronic Pelvic Pain. EAU Guidelines. Updated annually.
3. International Pelvic Pain Society (IPPS). Evaluation and Management of Chronic Pelvic Pain. Clinical Practice Resources.
4. Zondervan KT, Becker CM, Missmer SA. Endometriosis. New England Journal of Medicine. 2020;382:1244–1256.
5. As-Sanie S, Black R, Giudice LC, et al. Assessing Research Gaps and Unmet Needs in Endometriosis. American Journal of Obstetrics and Gynecology. 2019;221:86–94.
6. Morin M, Carroll MS, Bergeron S. Systematic Review of the Effectiveness of Physical Therapy Modalities in Women with Pelvic Floor Myofascial Pain and Dyspareunia. Sexual Medicine Reviews. 2017.
7. Jarrell J, Vilos G, Allaire C, et al. Consensus Guidelines for the Management of Chronic Pelvic Pain. Journal of Obstetrics and Gynaecology Canada. 2018.
8. Robert R, Prat-Pradal D, Labat JJ, et al. Anatomic Basis of Chronic Perineal Pain: Role of the Pudendal Nerve. Surgical and Radiologic Anatomy. 1998;20:93–98.
9. Labat JJ, Riant T, Robert R, et al. Diagnostic Criteria for Pudendal Neuralgia by Pudendal Nerve Entrapment (Nantes Criteria). Neurourology and Urodynamics. 2008;27:306–310.
10. Horne AW, Vincent K, Hewitt CA, et al. Gabapentin for Chronic Pelvic Pain in Women (GaPP2). The Lancet. 2020;396:909–917.
11. Younger J, Noor N, McCue R, Mackey S. Low-Dose Naltrexone for the Treatment of Fibromyalgia. Arthritis & Rheumatism. 2013;65:529–538.
12. Montenegro ML, Mateus-Vasconcelos EC, Rosa-e-Silva JC, et al. Importance of Pelvic Muscle Tenderness Evaluation in Women with Chronic Pelvic Pain. Pain Medicine. 2010.
13. Abbott JA. Gynecological Pain and the Role of Central Sensitization. Best Practice & Research Clinical Obstetrics and Gynaecology. 2014.
14. Aredo JV, Heyrana KJ, Karp BI, Shah JP, Stratton P. Relating Chronic Pelvic Pain and Endometriosis to Signs of Sensitization and Myofascial Dysfunction. Seminars in Reproductive Medicine. 2017.
15. Howard FM. Chronic Pelvic Pain. Obstetrics and Gynecology. 2003;101:594–611.
16. International Association for the Study of Pain (IASP). Terminology and Classification of Chronic Pain and Nociplastic Pain. IASP Clinical Resources.