Neuropathic Pain Explained: Diagnosis, Drug Treatment, Topical Therapies, and Practical Management

Neuropathic Pain: What It Is, Why It Matters, and How It Is Managed
Neuropathic pain is common, disabling, and clinically important because it behaves differently from nociceptive pain and usually responds to a different treatment framework. It arises from a lesion or disease of the somatosensory nervous system, is frequently associated with sleep disturbance, anxiety, depressed mood, and loss of function, and often requires a multimodal rather than single-treatment approach. The goal is rarely complete pain abolition. More often, good care means a meaningful reduction in pain together with better sleep, better tolerance of activity, and improved day-to-day function.
1. What Is Neuropathic Pain?
Neuropathic pain is defined by the International Association for the Study of Pain as pain caused by a lesion or disease of the somatosensory nervous system. That definition matters because it separates neuropathic pain from nociceptive pain, which arises from tissue injury or inflammation, and from nociplastic pain, which reflects altered nociceptive processing without clear evidence of tissue damage or somatosensory system lesion. In practice, this means neuropathic pain should not be diagnosed from descriptors alone. Burning, shooting, electric shock-like, tingling, or stabbing pain may strongly suggest neuropathic mechanisms, but the pain must still fit a plausible neuroanatomical pattern and a relevant disease process.
Neuropathic pain affects roughly 7–10% of the general population and a much higher proportion of people attending chronic pain services. Common causes include diabetic peripheral neuropathy, postherpetic neuralgia, painful radiculopathy, trigeminal neuralgia, chemotherapy-induced peripheral neuropathy, central post-stroke pain, spinal cord injury pain, post-surgical nerve injury, and small-fibre neuropathy. These are not interchangeable conditions, and one of the recurring clinical errors is to treat all “nerve pain” as if it were biologically identical.
Common causes of neuropathic pain
| Category | Condition | Somatosensory structure involved |
|---|---|---|
| Metabolic | Diabetic peripheral polyneuropathy | Peripheral sensory nerves |
| Infectious / post-infectious | Postherpetic neuralgia | Dorsal root ganglion and cutaneous nerves |
| Structural / compressive | Cervical or lumbar radiculopathy | Nerve root |
| Cranial nerve disorder | Trigeminal neuralgia | Cranial nerve V |
| Central | Central post-stroke pain | Spinothalamic / thalamic pathways |
| Central | Spinal cord injury pain | Spinal cord |
| Iatrogenic | Chemotherapy-induced peripheral neuropathy | Peripheral sensory nerves |
| Traumatic / surgical | Post-surgical or post-traumatic neuralgia | Peripheral nerves |
| Idiopathic / selective fibre loss | Small-fibre neuropathy | Unmyelinated C fibres |
2. How Neuropathic Pain Develops
Neuropathic pain develops through a combination of peripheral abnormal firing and central amplification. Peripherally, injured nerves and dorsal root ganglion neurons may generate ectopic discharges because of altered sodium-channel expression and lowered thresholds. Centrally, persistent afferent input can drive sensitisation in the dorsal horn, reduce inhibitory control, and recruit neuroinflammatory pathways involving glia and cytokines. The clinical consequence is that pain may continue long after the original lesion has stabilised, and the pain system becomes abnormally efficient at generating pain.
For clinical purposes, the key mechanistic themes are straightforward. Peripheral sensitisation helps explain burning pain, allodynia, and pain triggered by light touch. Central sensitisation helps explain spread, amplification, and exaggerated responses to relatively minor stimuli. Loss of inhibitory control helps explain why drugs that enhance descending noradrenergic and serotonergic inhibition, such as TCAs and SNRIs, can be effective.
The below figure has been adapted from N Engl J Med 2003;348:1243-1255 (Published March 27, 2003)
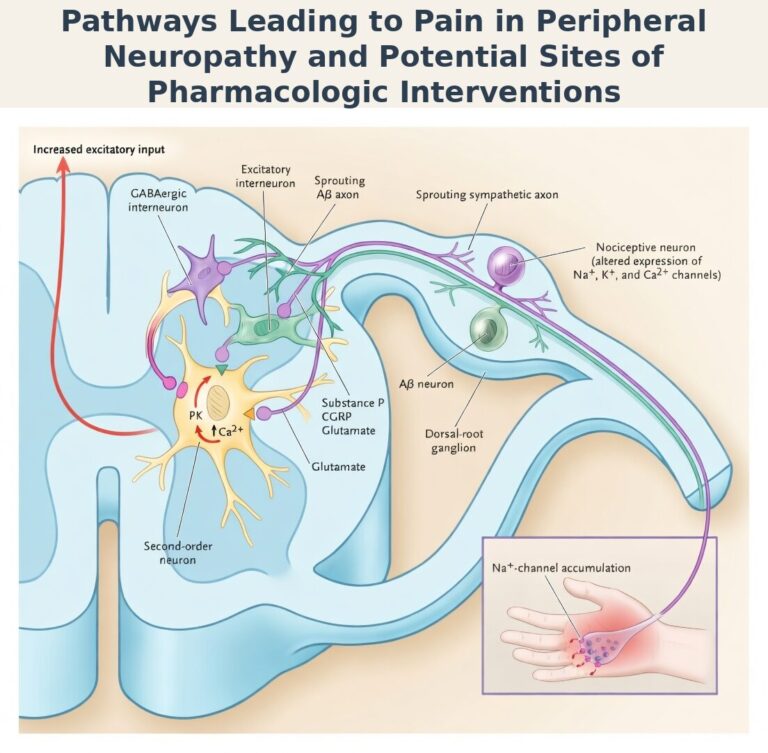
Sensory phenotypes and why they matter
| Phenotype | Typical features | Clinical implication |
|---|---|---|
| Irritable nociceptor | Burning pain, allodynia, hyperalgesia, relatively preserved sensation | Local treatments such as lidocaine, capsaicin 8%, or BTX-A may work particularly well |
| Deafferentation | Numbness with paradoxical burning, reduced sensory input, less allodynia | Systemic agents are often more relevant |
| Mixed phenotype | Background numbness with superimposed allodynia or spontaneous pain | Combination therapy is often required |
3. Clinical Presentation
Neuropathic pain has a characteristic clinical signature. Patients typically describe burning, electric shock-like, shooting, stabbing, lancinating, tingling, or pins-and-needles sensations. Spontaneous pain may be continuous, as in diabetic neuropathy or postherpetic neuralgia, or paroxysmal, as in trigeminal neuralgia. Evoked pain may include dynamic mechanical allodynia, punctate hyperalgesia, or cold allodynia. Negative sensory signs such as numbness, reduced pinprick, impaired thermal detection, and reduced vibration sense may coexist with pain and are often diagnostically helpful.
Typical evoked pain phenomena
| Type | Definition | Bedside example | Example conditions |
|---|---|---|---|
| Dynamic mechanical allodynia | Pain from light brushing or stroking | Cotton wool / soft brush | PHN, CRPS |
| Static mechanical allodynia | Pain from gentle sustained pressure | Finger pressure | PHN, diabetic neuropathy |
| Punctate hyperalgesia | Exaggerated pain to pinprick | Blunt pin | Post-surgical neuralgia |
| Cold allodynia | Pain from non-painful cold | Cool object / acetone | CRPS, small-fibre neuropathy |
| Heat hyperalgesia | Exaggerated pain to warmth | Thermal testing | Inflammatory neuropathy |
4. Diagnosis
The diagnosis of neuropathic pain is clinical and requires a structured assessment. No questionnaire is diagnostic on its own. The IASP grading system remains the most useful framework: the pain should lie in a plausible neuroanatomical distribution, there should be a history suggesting a relevant lesion or disease of the somatosensory system, sensory signs should support the diagnosis, and confirmatory testing should be used when necessary to achieve “definite” neuropathic pain.
IASP grading system
| Grade | Criteria required | Clinical meaning |
|---|---|---|
| Possible | History + plausible distribution | Neuropathic pain suspected |
| Probable | History + distribution + sensory signs | Strong clinical diagnosis |
| Definite | Above + confirmatory test | Objective confirmation of lesion or disease |
Screening tools such as DN4, PainDETECT, and LANSS are helpful because they improve recognition, especially in non-specialist settings, but they should be treated as screening aids rather than replacements for examination and clinical reasoning.
5. Diagnostic Algorithm for Peripheral Neuropathy
When peripheral neuropathic pain is suspected, the most useful sequence is history, examination, targeted blood tests, electrophysiology when large-fibre disease is suspected, and skin biopsy or quantitative sensory testing when small-fibre neuropathy is a concern. Imaging is added when central causes or structural lesions are suspected. This stepwise logic matters because it identifies treatable causes, prevents over-testing, and helps distinguish diffuse polyneuropathy from focal or central processes.
Stepwise diagnostic approach
| Step | Action | Reason |
|---|---|---|
| 1 | Characterise quality, distribution, onset, triggers, weakness, autonomic symptoms | Establish neuroanatomical plausibility |
| 2 | Examine sensation, reflexes, strength, vibration, temperature | Identify positive and negative signs |
| 3 | Blood tests: FBC, U&E, LFTs, HbA1c, glucose, B12 ± MMA, TFTs, SPEP | Find common treatable causes |
| 4 | NCS / EMG if large-fibre involvement suspected | Confirm and characterise neuropathy |
| 5 | Skin biopsy / QST if small-fibre neuropathy suspected | Detect disease missed by NCS |
| 6 | MRI if central or structural cause suspected | Rule out stroke, cord disease, root compression |
6. Principles of Management
Treatment should be realistic, structured, and multimodal. A clinically meaningful outcome is usually at least a 30% reduction in pain, but even when pain relief is incomplete, better sleep, better function, lower distress, and improved tolerance of activity all count as worthwhile gains. The most common mistake is to treat neuropathic pain as if one well-chosen drug should solve the problem. The evidence does not support that expectation. NNT values of 4.6 to 8.9 mean that most patients will not achieve dramatic pain relief from any single agent, which is why titration, switching, combination therapy, local treatments, and non-pharmacological support remain central.
7. First-Line Medications for Neuropathic Pain
This is the centrepiece of the article, because in routine practice the most important question is not simply “what helps neuropathic pain?” but which first-line drug is most appropriate for this patient, in this phenotype, with these comorbidities and risks? The three principal systemic first-line classes remain TCAs, SNRIs, and gabapentinoids. They have broadly comparable efficacy at population level, but they differ materially in tolerability, organ-function constraints, interaction burden, and practical clinical fit. The key principle is simple: choose the agent based on the patient, not the NNT alone.
First-line drug overview
| Class | Examples | Main mechanism | Best clinical fit | Key limitations |
|---|---|---|---|---|
| TCAs | Amitriptyline, nortriptyline | Enhance descending inhibition; sodium-channel effects | Pain with insomnia; younger patients without significant cardiac or anticholinergic risk | Dry mouth, constipation, sedation, falls, QT concerns |
| SNRIs | Duloxetine, venlafaxine | Noradrenergic / serotonergic descending inhibition | Pain with depression or anxiety; diabetic neuropathy | Nausea, BP effects, discontinuation symptoms, hepatic/renal cautions |
| Gabapentinoids | Gabapentin, pregabalin | α2δ calcium-channel modulation | Widespread neuropathic pain, pain with sleep disturbance or anxiety | Sedation, dizziness, oedema, falls risk, misuse potential |
NNT / NNH table for first-line decision-making
| Class | NNT | NNH | Clinical implication |
|---|---|---|---|
| TCAs | 4.6 | 17.1 | Strong efficacy signal, but tolerability limits use in many older or medically complex patients |
| SNRIs | 7.4 | 13.9 | Often attractive where mood symptoms coexist; duloxetine especially useful in diabetic neuropathy |
| Gabapentinoids | 8.9 | 26.2 | Slightly weaker efficacy signal but relatively favourable harm profile in carefully selected patients |
These values are valuable for shared decision-making. They show that none of these drug classes is universally dominant, and that modest benefit remains the norm rather than the exception.
How to choose between first-line drugs in real clinical practice
There is no universally best first-line drug. In practice, the decision is driven by comorbidity, phenotype, adverse-effect vulnerability, organ function, localisation of pain, and patient goals. Duloxetine is often attractive when depression, anxiety, or diabetic neuropathy coexist. Amitriptyline may be useful when sleep disturbance is prominent and the patient is younger and not burdened by cardiac or anticholinergic risks. Pregabalin or gabapentin may be reasonable where anxiety, hyperarousal, or widespread neuropathic pain are dominant, but they must be used more cautiously in frailty, renal impairment, gait instability, or patients already taking other sedatives. Frail older patients with localised peripheral neuropathic pain are often better served by topical lidocaine early, rather than by aggressive systemic escalation.
| Clinical scenario | Often favoured | Often avoided / used cautiously |
|---|---|---|
| Depression or significant anxiety | Duloxetine | TCA if anticholinergic burden is high |
| Prominent insomnia | Amitriptyline or nortriptyline | Avoid higher-dose TCA in frailty or cardiac disease |
| Frailty / older patient | Topical lidocaine, cautious duloxetine, cautious pregabalin | Amitriptyline |
| Cardiac disease / prolonged QT / conduction disease | Duloxetine or gabapentinoid may be safer | TCAs |
| Localised allodynic pain | Lidocaine plaster, capsaicin 8%, BTX-A later | Early broad systemic escalation |
| Widespread neuropathic pain | Systemic agent | Topicals alone |
| Trigeminal neuralgia | Carbamazepine / oxcarbazepine | Do not default to gabapentinoids as first choice |
Carbamazepine and trigeminal neuralgia
Trigeminal neuralgia remains an important exception to the broader first-line triad. Classical trigeminal neuralgia should not be managed as if it were interchangeable with diabetic neuropathy, PHN, or diffuse post-surgical neuropathic pain. Carbamazepine remains uniquely effective because its sodium-channel mechanism specifically targets the paroxysmal ectopic firing that characterises this condition. Oxcarbazepine is often used as a better-tolerated alternative.
8. How to Start Treatment Safely: Dosing Strategies and Monitoring
The most common reason first-line treatment appears to “fail” is not true pharmacological failure but inadequate dosing, inadequate duration, or premature discontinuation due to predictable side effects during titration. The safest and most effective principle remains: start low, go slow, but titrate adequately.
A useful clinical rule is that most patients should have an adequate trial at therapeutic or maximum tolerated dose for several weeks before a class is declared ineffective. Minimum effective dose thresholds matter. Gabapentin usually needs to reach at least 1200 mg/day, pregabalin at least 300 mg/day, duloxetine 60 mg/day, and venlafaxine at least 150 mg/day before the trial can really be judged.
Illustrative titration table
| Drug | Typical starting dose | Usual titration | Minimum effective target often worth reaching | Typical upper range |
|---|---|---|---|---|
| Amitriptyline | 10–25 mg at night | Increase every 5–7 days as tolerated | 25–50 mg | 75 mg in most pain practice |
| Nortriptyline | 10–25 mg at night | Increase every 5–7 days | 25–50 mg | 75 mg |
| Duloxetine | 20–30 mg/day | Increase after 1–2 weeks | 60 mg | 120 mg in selected patients |
| Venlafaxine | 37.5 mg/day | Increase gradually every few days | 150 mg | 225 mg |
| Gabapentin | 100–300 mg at night | Increase every 3 days in divided doses | 1200 mg/day | 3600 mg/day |
| Pregabalin | 25–75 mg at night or BD | Increase every few days | 300 mg/day | 600 mg/day |
TCA monitoring table
| Parameter | Why it matters | Practical action |
|---|---|---|
| ECG where relevant | QT prolongation / conduction concerns | Baseline ECG in older patients or cardiac risk; avoid QT stacking |
| Orthostatic BP | Falls / dizziness | Check during titration, especially in frailty |
| Sedation / cognitive burden | Falls, confusion | Night dosing, slower titration, avoid in frail elderly if problematic |
| Constipation / urinary retention | Anticholinergic burden | Ask proactively; avoid in BPH / retention risk |
| Dry mouth / blurred vision | Common tolerability issues | Counsel early; switch if burdensome |
SNRI monitoring table
| Parameter | Why it matters | Practical action |
|---|---|---|
| Blood pressure | Venlafaxine and sometimes duloxetine can affect BP | Baseline and periodic monitoring |
| Sodium where indicated | Hyponatraemia risk in susceptible patients | Consider in older adults or symptomatic patients |
| Renal function | Dose suitability and accumulation risk | Review before initiation and during treatment when relevant |
| Liver function for duloxetine | Hepatic risk | Avoid duloxetine in chronic liver disease |
| Discontinuation symptoms | Venlafaxine especially | Taper gradually; avoid abrupt stopping |
Gabapentinoid monitoring table
| Parameter | Why it matters | Practical action |
|---|---|---|
| Renal function | Renal clearance; accumulation risk | Dose-adjust early and reassess if renal function changes |
| Sedation / dizziness | Falls and cognitive slowing | Slow titration; warn patients early |
| Gait instability / falls | Especially relevant in older adults | Review balance and function at follow-up |
| Oedema / weight gain | Common practical adverse effects | Reassess if problematic, especially in frailty |
| Mood / suicidality | Class warning | Monitor if psychiatric vulnerability present |
| Misuse potential | Increasing concern in some populations | Use cautiously in substance-use risk |
9. Dose Adjustments and Precautions in Special Situations
This section needs to be clinician-facing because neuropathic pain prescribing often fails at the point of comorbidity rather than at the point of theoretical efficacy. Renal impairment materially increases gabapentinoid burden. Hepatic impairment changes SNRI suitability. Cardiac disease changes TCA safety. Frailty changes the balance between benefit and harm even when the pharmacology looks acceptable on paper.
| Clinical situation | Main concern | Most relevant drugs | Practical implication |
|---|---|---|---|
| Renal impairment | Drug accumulation, sedation, falls, respiratory risk when combined with opioids | Gabapentin, pregabalin | Reduce dose early and titrate more slowly; reassess if renal function worsens |
| End-stage renal disease / dialysis | Marked accumulation and higher toxicity risk | Gabapentin, pregabalin | Use very cautiously; avoid aggressive titration; combination with opioids is particularly risky |
| Hepatic impairment | Increased exposure and hepatotoxicity risk | Duloxetine, venlafaxine | Avoid duloxetine in significant chronic liver disease; use venlafaxine cautiously |
| Cardiac disease / prolonged QT / conduction abnormality | Arrhythmia and conduction risk | TCAs | Avoid or use with ECG-based caution; avoid combining with other QT-prolonging drugs |
| Older adults / frailty | Sedation, orthostasis, falls, cognitive burden, anticholinergic effects | TCAs, gabapentinoids | Start lower, titrate slower, and consider topical treatments earlier |
| Polypharmacy | Serotonergic toxicity, QT stacking, additive CNS depression | TCAs, SNRIs, tramadol, opioids | Check interactions carefully before combining agents |
| History of substance misuse | Misuse, dependence, escalating dose pressure | Pregabalin, gabapentin, opioids | Prescribe cautiously, review regularly, avoid risky combinations |
| Severe constipation / urinary retention / glaucoma / BPH | Worsening anticholinergic adverse effects | TCAs | Usually avoid tertiary amine TCAs |
| Marked anxiety / insomnia | May influence drug choice rather than dose adjustment | Pregabalin, duloxetine, amitriptyline | Choose according to overall symptom pattern rather than pain alone |
| Localised neuropathic pain in medically complex patient | Systemic adverse-effect burden may outweigh benefit | All systemic agents | Consider lidocaine plasters or capsaicin earlier |
In practical terms, gabapentin and pregabalin can accumulate substantially in renal dysfunction. Duloxetine should generally be avoided in chronic liver disease or significant hepatic impairment. TCAs require the greatest caution in cardiac disease because of QT prolongation and conduction concerns. Frailty often shifts the balance in favour of topical therapies sooner than one might otherwise choose in a younger and fitter patient.
10. How to Recognise Treatment Failure
Treatment failure needs a practical definition. The article should distinguish between true non-response, partial response, intolerance, and pseudo-failure due to inadequate trial. An adequate trial usually means reaching a therapeutic or maximum tolerated dose and maintaining it long enough to judge benefit properly.
A responder is a patient with clinically meaningful pain reduction, usually at least 30%, ideally accompanied by functional improvement. A partial responder is a patient with some meaningful benefit but inadequate overall control. A non-responder is a patient with no meaningful improvement despite adequate dose and duration. Intolerance means side effects prevent meaningful escalation or continuation. These distinctions matter because the management response differs in each situation.
A patient on gabapentin 300 mg at night who says it “did not work” has not had a fair trial. A patient on duloxetine 60 mg for six weeks with clearly improved mood and sleep but persistent pain may not have failed; they may be a partial responder and a candidate for intelligent add-on therapy. A patient who develops severe sedation, falls, or urinary retention early in titration has not “failed neuropathic pain treatment”; they have failed that particular drug choice.
11. What to Do When First-Line Treatment Fails
When first-line treatment fails, the key decision is whether to switch or add. The broad clinical rule is simple. If the first drug produced no meaningful benefit or caused unacceptable adverse effects, switching class is usually the right move. If it produced partial benefit, especially with some gains in sleep or function, add-on treatment may be more rational than abandoning the initial benefit.
This section is important because “failure” is often handled too crudely. Escalating a poorly tolerated agent is a mistake. Abandoning a partially helpful agent without considering complementary add-on treatment is also often a mistake. A structured progression makes more sense: confirm adequate dosing, decide whether the patient is a partial responder or true non-responder, and then either switch to a mechanistically different class or add a second agent if the first has provided some meaningful benefit.
| Clinical situation | Most sensible next step |
|---|---|
| No benefit and poor tolerability | Switch class |
| No benefit despite adequate dose and duration | Switch class and review diagnosis / phenotype |
| Partial benefit with acceptable tolerability | Add a second mechanistically different agent |
| Localised pain with systemic side effects | Move toward topicals, BTX-A, or local strategies |
| Frail patient accumulating side effects | De-escalate systemic burden and use local / non-drug options earlier |
Cross-tapering should be done thoughtfully when moving between classes, especially where abrupt discontinuation may cause withdrawal symptoms or loss of partial benefit. Particular caution is needed with serotonergic agents, and casual overlap of TCA and SNRI is generally best avoided because of serotonin-toxicity risk.
12. Combination Therapy When Monotherapy Is Not Enough
Combination therapy is important, but it should be used intelligently rather than reflexively. The best-supported model is sequential combination therapy: start with monotherapy, optimise it, and only if relief is suboptimal add a second agent from a different mechanistic class.
The practical message from OPTION-DM is not that combinations are dramatically superior in all cases. It is that monotherapy is often insufficient, and that partial responders may gain meaningful additional benefit from a second agent. The trial also showed that combinations involving pregabalin with either amitriptyline or duloxetine achieved similar efficacy overall, with some suggestion that pregabalin followed by amitriptyline had fewer discontinuations.
| Combination | Why it makes sense | Clinical comment |
|---|---|---|
| Gabapentinoid + TCA | Complementary mechanisms; useful when sleep disturbance coexists | Good option for partial responders if tolerated |
| Gabapentinoid + SNRI | Common practical pairing; useful in diffuse pain or diabetic neuropathy | Reasonable when duloxetine has helped partly |
| TCA + SNRI | Limited evidence and serotonin-toxicity concern | Generally avoid |
Combination therapy should therefore be framed as a monitored, individualised next step rather than a guaranteed improvement. A sensible sequence is to optimise first-line monotherapy, define whether the response is partial or absent, add a second agent from a different class if benefit is partial and tolerability is acceptable, titrate the second drug slowly while maintaining the first, and then reassess after an additional 6–10 weeks.
13. Why Gabapentinoid and Opioid Combinations Are Now Discouraged
This section needs to be explicit because the old rationale of “opioid-sparing” no longer justifies routine use of this combination. Historically, gabapentinoids were seen as attractive co-analgesics that might reduce opioid dose requirements while offering multi-mechanism pain control. The harm signal did not emerge from short randomised trials; it emerged later from population-level data and from better understanding of the pharmacodynamic interaction.
The crucial point is that this is not primarily a pharmacokinetic interaction. Gabapentinoids do not meaningfully alter opioid concentrations. The problem is pharmacodynamic: both drug classes suppress respiration through different mechanisms, and gabapentinoids appear capable of reversing opioid respiratory tolerance. That means a patient who has been “stable” on opioids may become newly vulnerable to respiratory depression when a gabapentinoid is added or up-titrated.
The earlier opioid-sparing logic focused only on analgesia and ignored mortality. Current evidence has shifted the balance decisively. Opioid-sparing is no longer, by itself, a sufficient justification for routine combination.
| Combination | Risk signal |
|---|---|
| Gabapentinoid + opioid | Approximately 2× fatal overdose risk vs opioid alone |
| Gabapentinoid + opioid + benzodiazepine | Highest-risk combination; markedly increased overdose and respiratory-depression risk |
| Pregabalin + opioid | Often worse than gabapentin in overdose datasets |
| ESRD / dialysis + gabapentinoid + opioid | Especially high risk because of accumulation |
If there is a rare situation in which the combination cannot be avoided, the safest approach is to use the lowest possible doses, titrate slowly, avoid triple therapy wherever possible, consider naloxone co-prescription, and monitor closely for sedation and respiratory compromise.
14. Topical Treatments for Localised Neuropathic Pain
Topicals deserve more weight than they often receive, particularly in localised peripheral neuropathic pain and in patients where systemic burden matters. They are not merely fallback options. In frail or elderly patients, lidocaine 5% patch may effectively function as a first-line treatment.
Lidocaine 5% plasters
Lidocaine plasters are particularly attractive for clearly localised peripheral neuropathic pain, especially when allodynia is present and the painful area can be mapped. Their main strength is not dramatic efficacy in every patient, but excellent tolerability and very low systemic burden. That makes them especially useful in frailty, polypharmacy, renal impairment, and older patients who would be at higher risk from oral agents. Practical use usually involves wearing the plaster for a fixed daily period rather than continuously. They are much less useful for diffuse neuropathic pain, where the biology and pain distribution simply exceed what a local patch can reasonably treat.
Capsaicin 8% patch
Capsaicin 8% patch is best thought of as a specialist, phenotype-sensitive topical treatment for localised peripheral neuropathic pain. Its mechanism is not simple “numbing.” It acts through intense activation and subsequent defunctionalisation of TRPV1-expressing nociceptors. In practical terms, it is most relevant where there is preserved nociceptor function, such as the irritable nociceptor phenotype with allodynia or hyperalgesia. Initial application discomfort or burning is expected and should be explained in advance. Its advantage is that a single treatment can provide benefit over a prolonged period, rather than requiring daily application.
Low-concentration capsaicin cream and lidocaine 5% cream
Low-concentration capsaicin cream remains an option when capsaicin 8% patch is unavailable, but it has weaker evidence and lower convenience because repeated applications are required. Lidocaine 5% cream can be discussed as a more flexible local anaesthetic option in selected patients, but the evidence base is stronger for lidocaine plasters than for cream, and that hierarchy should be respected.
| Topical | Best use case | Main advantage | Main limitation |
|---|---|---|---|
| Lidocaine 5% plaster | Localised peripheral neuropathic pain, frailty, polypharmacy | Very low systemic burden | Less useful for widespread pain |
| Capsaicin 8% patch | Localised pain with preserved nociceptor function / allodynia | Longer-lasting effect after single application | Initial application discomfort |
| Low-concentration capsaicin cream | When 8% patch unavailable | Accessible option | Repeated application; weaker evidence |
| Lidocaine 5% cream | Selected localised use | Flexible application | Less robust evidence than plasters |
15. Compounded Topical Treatments: Do They Have a Role?
Compounded topical treatments are often discussed for refractory localised neuropathic pain, but they are not first-line, not routine second-line, and should usually be reserved for patients who have already failed approved topicals and appropriate systemic therapy. The largest randomised trial found that compounded creams were not better than placebo for localised chronic pain, including neuropathic pain, which should significantly temper routine use.
| Agent | Typical concentration | Proposed mechanism | Evidence quality | Practical message |
|---|---|---|---|---|
| Ketamine | 5–10% | NMDA antagonism | Very low | No convincing evidence as a standalone topical strategy |
| Amitriptyline | 2–5% | Sodium-channel and monoaminergic effects | Low / conflicting | Marginal benefit at best |
| Gabapentin | 5–6% | α2δ modulation | Very low | No robust RCT evidence |
| Baclofen | 2–5% | GABA-B agonism | Very low | Usually appears only in mixed preparations |
| Mixed creams | Variable | Multi-target | Low | Largest RCT showed no meaningful benefit over placebo |
The practical niche for compounded topicals is therefore limited. They may occasionally be considered when pain is clearly localised, the patient cannot tolerate systemic therapy, approved topicals have failed, and the patient understands that benefit is uncertain and costs may be out-of-pocket.
16. Botulinum Toxin Type A for Refractory Localised Neuropathic Pain
Botulinum toxin A deserves a separate section because it is a stronger local option than compounded topicals for selected refractory patients. It appears most useful in localised peripheral neuropathic pain with allodynia, especially where an irritable nociceptor phenotype is preserved. Its appeal is obvious in the right patient: no systemic adverse-effect burden, repeatability, and potentially durable benefit over around three months.
In clinical practice it should be thought of as a serious option for patients who have failed or not tolerated first-line oral agents and approved topical therapies, rather than as a fringe treatment. It is often reasonable to give at least two treatment sessions before declaring failure.
17. Non-Pharmacological Approaches
Non-pharmacological care is not optional. CBT, structured exercise, education, and in selected refractory cases spinal cord stimulation are evidence-based parts of care rather than last-resort extras.
In practical terms, four messages matter. First, education and reassurance matter because uncertainty amplifies pain and fuels health anxiety. Second, activity restoration and pacing matter because fear-avoidance and deconditioning can become major drivers of disability even when pain remains biologically neuropathic. Third, psychological strategies such as CBT can reduce distress, catastrophising, sleep disruption, and the sense of helplessness that often accompanies persistent neuropathic pain. Fourth, TENS, desensitisation, and multidisciplinary pain management can be particularly useful when systemic drug tolerance is poor or the patient is stuck between partial benefit and unacceptable side effects.
18. Neuromodulation and Specialist Options for Refractory Neuropathic Pain
For refractory neuropathic pain, especially painful diabetic neuropathy, specialist options such as rTMS and spinal cord stimulation deserve inclusion because they complete the treatment pathway. These therapies are not early-line treatments, but they matter because they change the conversation from “nothing else works” to “there are structured specialist options when standard measures have been exhausted.”
19. Treatments That Are Less Useful or Generally Not Recommended
❌ Levetiracetam — strong recommendation against use because trials have generally been negative.
❌ Mexiletine — strong recommendation against because of limited efficacy and safety concerns.
❌ Cannabinoids — weak recommendation against in current guideline positioning.
❌ Valproate — weak recommendation against in routine neuropathic pain practice.
❌ SSRIs as first-line analgesics — not recommended as first-line neuropathic pain agents when better-supported alternatives exist.
❌ Routine opioid escalation — no longer appropriate as a default strategy for chronic neuropathic pain.
20. A Practical Stepwise Treatment Algorithm
History, neuroanatomical distribution, sensory signs, and appropriate investigations where needed.
Localised vs widespread pain, irritable nociceptor vs deafferentation phenotype, comorbidities, frailty, sleep and mood disturbance.
TCA, SNRI, gabapentinoid, or in selected localised / frail patients, topical therapy early.
Start low, go slow, but reach a meaningful therapeutic dose where tolerated.
Responder, partial responder, non-responder, or intolerance.
Switch class.
Consider combination therapy.
Consider lidocaine, capsaicin 8%, or BTX-A.
Do not routinely combine gabapentinoids with opioids simply for “opioid-sparing”.
Multidisciplinary pain management, rTMS, spinal cord stimulation, specialist review.
21. Key Take-Home Messages
- Neuropathic pain is a distinct clinical entity and should not be diagnosed from pain descriptors alone.
- Accurate diagnosis depends on neuroanatomical plausibility, clinical examination, and appropriate investigation where required.
- TCAs, SNRIs, and gabapentinoids remain the main systemic first-line options, but there is no universally best drug.
- Drug choice should be guided by phenotype, comorbidities, organ function, frailty, localisation of pain, and patient goals.
- Topical treatments are particularly important in localised peripheral neuropathic pain and may be especially useful in frail or polymedicated patients.
- Many apparent treatment failures are actually failures of dose, duration, or drug selection, rather than proof that neuropathic pain is untreatable.
- Partial responders often benefit from rational combination therapy, particularly gabapentinoid plus TCA or gabapentinoid plus SNRI.
- Gabapentinoid-opioid combinations should no longer be viewed as routine “opioid-sparing” treatment, because the safety risks now outweigh that older logic in most patients.
- Non-pharmacological management is essential, including education, pacing, rehabilitation, and psychological support where appropriate.
- The goal is not always complete pain relief, but meaningful improvement in pain, sleep, function, and quality of life.
22. References
Soliman N, Moisset X, Ferraro MC, et al. Pharmacotherapy and Non-Invasive Neuromodulation for Neuropathic Pain: A Systematic Review and Meta-Analysis. Lancet Neurology. 2025.
Mauermann ML, Staff NP. Peripheral Neuropathy. JAMA. 2026.
Tesfaye S, Sloan G, Petrie J, et al. OPTION-DM. Lancet. 2022.
Price R, Smith D, Franklin G, et al. AAN Practice Guideline Update: Oral and Topical Treatment of Painful Diabetic Polyneuropathy. Neurology. 2022.
Additional topical, BTX-A, and safety references are reflected in the uploaded source documents used to build this draft.