Gabapentin and Pregabalin in Persistent Pain: Evidence, Risks, and Safer Long-Term Strategies

Gabapentinoids in Persistent Neuropathic Pain: Benefits, Risks, and Safer Long-Term Strategies
Gabapentinoids—gabapentin and pregabalin—are widely used for neuropathic pain and have often been viewed as “safer” alternatives to opioid analgesics. Over the past decade, however, growing real-world evidence has raised concerns about long-term adverse effects (including possible cognitive risks), tolerance and dependence, misuse/diversion, and particularly dangerous outcomes when combined with opioids and other sedatives.
This article reviews the evolving evidence on harms and limitations of long-term gabapentinoid prescribing, explains why medication-only strategies are frequently not the best solution for persistent pain, and outlines how carefully selected interventional pain treatments—performed with modern imaging guidance—can help some patients reduce pain, improve function, and decrease reliance on long-term medication.
What Are Gabapentinoids and Why Are They Used?
Gabapentin and pregabalin bind to the α2δ subunit of voltage-gated calcium channels in the nervous system. This reduces release of excitatory neurotransmitters and can dampen neuronal hyperexcitability—one mechanism contributing to neuropathic pain. In the UK, they are licensed for neuropathic pain and as adjunct therapy for epilepsy; pregabalin is also licensed for generalised anxiety disorder.
For some patients, gabapentinoids reduce neuropathic pain intensity and improve sleep. But at a population level, only a minority achieve sustained, clinically meaningful benefit—particularly when these medicines are used outside clear neuropathic pain indications.
Because persistent pain is often multi-factorial, it is common for patients to remain on these medicines long term despite limited benefit, while accumulating adverse effects and interacting medications.
Emerging Evidence: Cognitive Impairment and Possible Dementia Risk
Recent observational studies have reported an association between long-term gabapentinoid exposure and higher rates of cognitive impairment and dementia diagnoses in some populations. These studies do not prove causation, but the consistency of the signal—along with plausible biological mechanisms and patient-reported cognitive symptoms—means the risk should be taken seriously in long-term prescribing decisions.
Clinically, many patients describe cognitive effects well before any formal diagnosis: “brain fog,” reduced concentration, memory lapses, slowed processing speed, and impaired multitasking. These effects can influence safety (driving, falls risk), employment, and quality of life—particularly when combined with other centrally acting drugs or in patients with sleep disturbance, depression, and comorbidity.
A sensible approach is to treat gabapentinoids as time-limited trials with clear goals, and to monitor cognition, sedation, and function if longer-term use is considered.
Prescribing Trends in the UK: What the Data Really Show
Large-scale UK primary care analyses demonstrate how dramatically gabapentinoid prescribing rose over the last two decades. In national data from the Clinical Practice Research Datalink, incident gabapentin prescribing increased after approval and peaked in 2016–17, while incident pregabalin prescribing peaked later in 2017–18. Since then, new prescribing has begun to fall modestly, particularly for gabapentin, but prevalent prescribing has largely plateaued—suggesting many established users continue these medicines long term.
The same UK primary care data highlight extensive polypharmacy: gabapentinoids are frequently co-prescribed with other centrally acting medications, including opioids, antidepressants, benzodiazepines, and Z-drugs. A key concern is that a substantial proportion of patients do not have a clearly recorded licensed indication, and chronic back pain features prominently around the time of initiation in many cases—conditions where long-term benefit is uncertain.
Although gabapentin and pregabalin were reclassified as controlled drugs in 2019, the immediate impact on long-term (prevalent) prescribing appears limited—illustrating the difficulty of deprescribing once patients become established on these medicines and the need for safer, more effective alternatives for persistent pain.

Gabapentinoids and Opioids: A High-Risk Combination
Prescribing data from around 2017–2018 suggest that approximately 3% of adults in England received at least one gabapentinoid prescription (gabapentin or pregabalin), while around 13% received an opioid prescription for chronic non-cancer pain. These figures reflect prescribed use only and do not capture illicit consumption, which has become an important contributor to harm.
Co-prescribing is common: depending on the patient cohort and how overlap is defined, studies report that 15% to 70% of people prescribed gabapentinoids are also prescribed opioids. Scottish data from Tayside and Fife (2016) found that nearly half of recurrent gabapentinoid users were also co-prescribed an opioid—illustrating how frequently this combination occurs in real-world practice.
This matters because both gabapentinoids and opioids depress the central nervous system. When used together, their effects can be additive, increasing the risk of profound sedation, impaired cognition, falls, respiratory depression, accidental overdose, and death. UK drug-related deaths involving gabapentinoids rose sharply between 2012 and 2016, and concerns about misuse, diversion, and fatal combinations contributed to their reclassification as controlled drugs in 2019.
Prescribing rates for both drug classes are generally higher in women, older adults, and people living in areas of greater socioeconomic deprivation—groups who may also face higher burdens of comorbidity and increased vulnerability to medication-related harm.

Other Long-Term Adverse Effects: What Patients Commonly Experience
Beyond cognitive concerns, gabapentinoids are associated with adverse effects that can become more clinically significant with time, dose escalation, and concurrent sedatives. Common problems include dizziness, drowsiness, fatigue, blurred vision, peripheral oedema, weight gain, dry mouth, and gait instability—factors that increase falls and injury risk, particularly in older adults.
There is also growing recognition of tolerance and dependence. Some patients experience diminishing benefit after an initial response, leading to upward titration without proportional pain relief. When reduced or stopped abruptly, withdrawal symptoms can occur (anxiety, insomnia, nausea, sweating, irritability, tremor, and rebound pain), making deprescribing difficult unless it is planned and supported.
Misuse and diversion are now well recognised. Most patients use these medicines appropriately, but euphoric or calming effects reported by some individuals mean there is misuse potential, particularly in vulnerable populations or those with a history of substance misuse. Safe prescribing therefore requires careful assessment, informed consent, time-limited trials, and regular review of benefit versus harm.
Why Medication-Only Management Is Often Not the Best Long-Term Strategy
Persistent pain is rarely a single “pain signal” that can be switched off with a tablet. It is influenced by peripheral pathology, central sensitisation, sleep disruption, mood and stress responses, deconditioning, and social context. Medications that target a single mechanism may help some patients, but they seldom address the broader drivers of chronic pain.
Over time, medication-centric strategies can expose patients to cumulative harms: sedation, cognitive impairment, falls risk, weight gain, dependence, and dangerous drug interactions. Polypharmacy can also unintentionally reinforce passive coping (“the pill will fix it”) while delaying more effective strategies aimed at restoring function, confidence, and quality of life.
For these reasons, long-term pain care often works best when medications are used as time-limited tools within a broader plan—focused on function, activity, sleep, psychological wellbeing, and targeted treatments aimed at the underlying pain generator where possible.
How Interventional Pain Treatments Can Help
Interventional pain management can be valuable for carefully selected patients—particularly where a specific pain generator can be identified. Treatments such as targeted nerve blocks, radiofrequency procedures, and image-guided injections aim to disrupt pathological pain signalling closer to its source rather than broadly suppressing the nervous system.
For the right patient, successful intervention can reduce pain intensity, improve mobility and sleep, and help reduce reliance on long-term medication. This can be particularly important when medication side effects, tolerance, or high-risk combinations (such as gabapentinoids with opioids) are limiting safe prescribing options.
Limitations of Interventional Treatments (And Why Patient Selection Matters)
It is important to be honest and balanced: interventional treatments are not suitable for everyone and are not a cure-all for persistent pain. Outcomes depend on accurate diagnosis, careful patient selection, and technical precision. If the pain driver is not clearly identified—or if central sensitisation is the dominant mechanism—procedures may offer limited benefit.
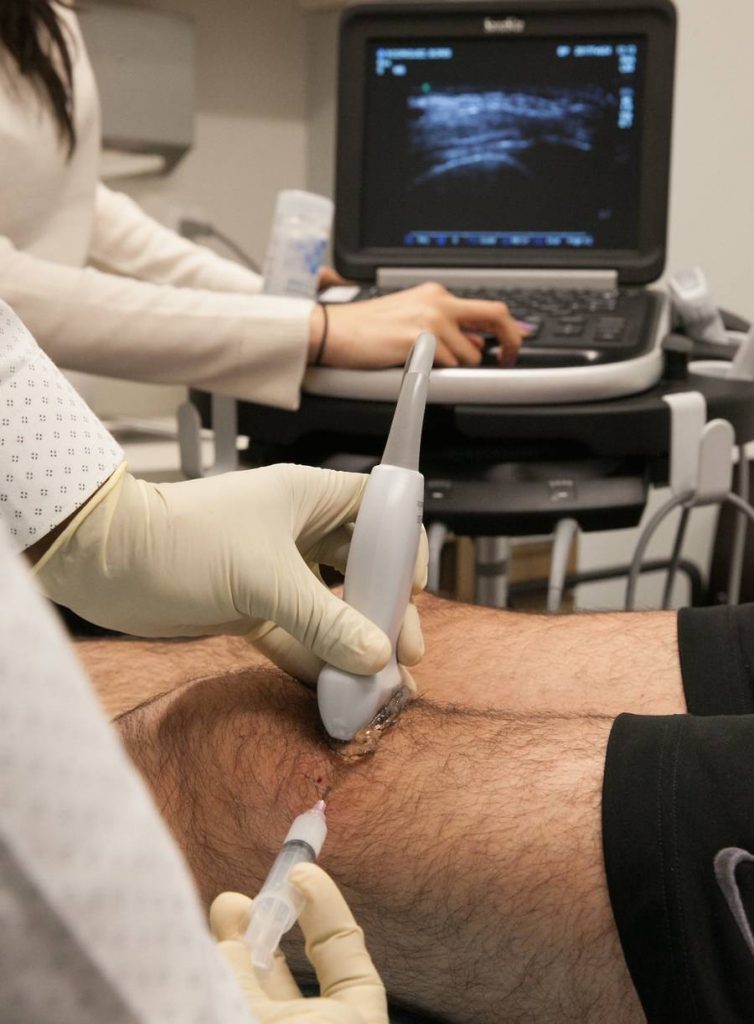
However, if patients are chosen correctly and appropriately, and treatments are performed by experts using modern real-time ultrasound guidance, results can be spectacular. Ultrasound guidance improves precision and safety by allowing the clinician to visualise target structures, avoid vulnerable anatomy, and place medication or perform procedures with greater accuracy.
As with any treatment, realistic expectations, shared decision-making, and outcome monitoring are essential. The aim is improved function and quality of life—not simply “a number” on a pain scale.
Interventional Pain Care at Pain Spa
At Pain Spa, Dr Krishna has extensive experience delivering interventional pain treatments for patients with persistent pain. This includes diagnostic blocks and therapeutic procedures such as ultrasound-guided injections and radiofrequency-based treatments, tailored to the individual’s pain pattern and clinical assessment.
Care is focused on accurate diagnosis, careful patient selection, and performing treatments with precision under ultrasound guidance. This approach aims to maximise outcomes while minimising risk, and is often integrated with broader rehabilitation and medication review strategies to support longer-term improvement.
For patients who have struggled with medication side effects or who are exposed to high-risk drug combinations, a targeted interventional approach—where clinically appropriate—may offer a pathway to better pain control, improved function, and reduced reliance on long-term pharmacotherapy.
A Balanced, Individualised Approach
Gabapentinoids still have a role in neuropathic pain management, particularly as short, structured trials with clear goals and regular review. Long-term use should trigger active monitoring for sedation, falls risk, cognitive symptoms, weight change, and signs of dependence—especially when combined with opioids or other sedatives.
For many people with persistent pain, the best outcomes come from integrating the right options at the right time: supported self-management, rehabilitation, sleep and mental health optimisation, careful medication review, and—where appropriate—interventional pain treatments performed with modern imaging guidance.
The goal is not to demonise medication or suggest procedures work for everyone. The goal is safer, more effective long-term pain care that prioritises function, independence, and quality of life.