Opioids in Persistent Pain: Evidence, Risks, and Why Long-Term Use Often Fails

Opioids in Chronic (Persistent) Pain: Limitations, Risks, and Safer Long-Term Strategies
Opioids can be very effective for acute pain, cancer pain, and end-of-life care. However, their role in long-term non-cancer persistent pain is far more limited than many people expect. Over time, the balance between benefit and harm often shifts in the wrong direction—especially as doses escalate or opioids are combined with other sedating medicines.
This article explains why opioids are generally not recommended as a routine long-term solution for persistent non-cancer pain, summarises the clinical evidence, outlines the risks of dependence and addiction, and provides practical, patient-centred strategies for safe prescribing and opioid deprescribing. It also clarifies common misconceptions—particularly around opioid patches and fentanyl—and explains how specialist support can help patients reduce opioids safely without automatically worsening pain.
Why Opioids Became So Common in Non-Cancer Pain
Persistent pain is common, disabling, and complex. In many healthcare systems, opioid prescribing expanded rapidly for non-cancer pain over the last two decades, driven by high unmet need, limited access to multidisciplinary pain services, time pressures in primary care, and early optimism that long-term opioid therapy would maintain function and quality of life.
Public health reviews have also highlighted how wider system influences—including prescribing culture, availability of potent formulations, and commercial promotion in some settings—contributed to increased opioid exposure at a population level. This matters because increased prescribing has been accompanied by rising opioid burden (overall “opioid load”), higher rates of diversion and misuse, and preventable harms.
The key clinical reality is that many people remain on opioids for years despite only modest pain improvement and little sustained functional gain.
Graphs: Opioid Prescribing and Opioid Burden Over Time
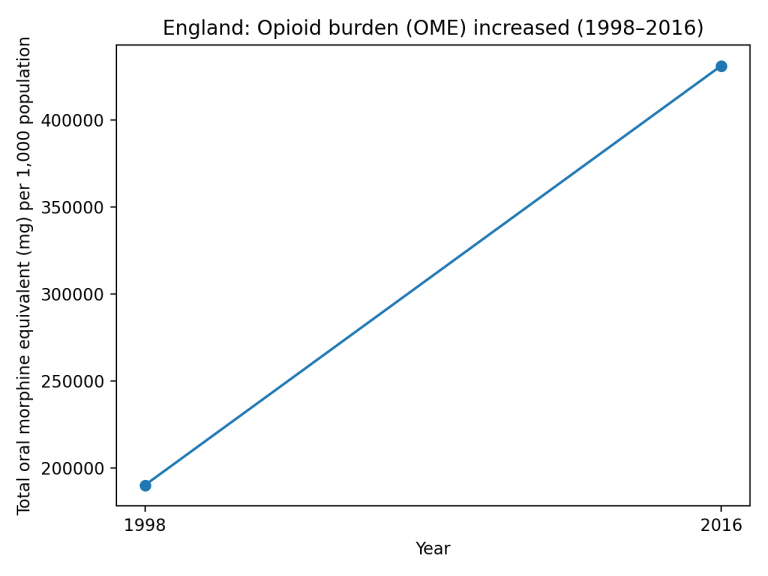
Rise in opioid prescribing and the increase in total oral morphine equivalent (OME) burden over time
Figure 1. England: Opioid prescriptions per 1,000 patients (1998–2016).
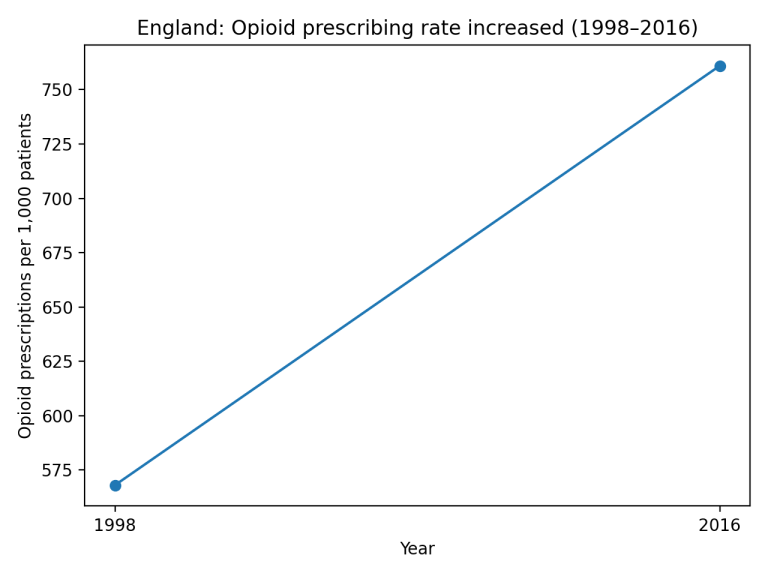
Figure 2. England: Total oral morphine equivalent (OME) burden per 1,000 population (1998–2016).
What Does the Evidence Show for Chronic (Non-Cancer) Pain?
For chronic non-cancer pain, the best available evidence suggests opioids can provide small improvements in pain intensity in the short-to-medium term, but evidence for sustained benefit beyond 6–12 months is limited. Even when pain scores improve slightly, improvements in physical function and quality of life are often small, inconsistent, or absent.
At the same time, risks tend to accumulate with longer duration and higher dose, including overdose, fractures and falls, endocrine dysfunction, constipation, cardiovascular events, and opioid use disorder.
Limitations of Long-Term Opioid Therapy
1) Tolerance and dose escalation: With repeated exposure, tolerance to analgesic effects commonly develops. This can lead to higher doses being needed to achieve the same effect, which increases risk without guaranteeing better pain control.
2) Opioid-induced hyperalgesia: In some patients, long-term opioid exposure can paradoxically increase pain sensitivity. In this situation, increasing the opioid dose can worsen pain rather than improve it; dose reduction may be the more effective strategy.
3) Long-term systemic harms: Harms may include sedation, cognitive impairment, falls and fractures, constipation and gastrointestinal complications, endocrine dysfunction (including opioid-induced androgen deficiency), immune effects, and cardiovascular events—many of which are dose-dependent.
4) Medication interactions: Combining opioids with other sedating medicines (e.g., benzodiazepines, Z-drugs (non-benzodiazepine hypnotic medications), gabapentinoids, alcohol) increases the risk of respiratory depression, overdose, and death.
Dependence, Addiction, and Why “Therapeutic Use” Is Not Risk-Free
Physical dependence means the body adapts to the medicine, so withdrawal symptoms can occur if opioids are stopped suddenly. This is expected with long-term use and is not the same as addiction.
Addiction (opioid use disorder) is a separate clinical condition characterised by impaired control, compulsive use, craving, and continued use despite harm. It develops in a minority of patients, but risk increases with higher doses, longer duration, comorbid mental health conditions, and concurrent sedatives.
One important safety issue is that tolerance to analgesia can develop while the risk of respiratory depression persists. This means dose escalation—whether prescriber-driven or patient-driven—can increase overdose risk even when the intention is simply to control pain.
Safe and Responsible Prescribing: What “Good Practice” Looks Like
Where opioids are used for persistent pain, safe prescribing requires clear goals, regular review, and an agreed plan for stopping if meaningful benefit is not achieved. Core principles include: the lowest avoiding ineffective dose escalation; preference for short-acting use in initial trials; avoidance of high-risk combinations; monitoring for sedation, falls, mood and cognitive change; and careful review of function and quality of life—not just pain scores.
A key governance principle is to agree an “end-of-treatment” plan at the start, because the risk of dependence and withdrawal rises sharply beyond about 3 months of use.
The Opioid Epidemic: A Cautionary Lesson
The opioid epidemic demonstrates how quickly well-intentioned prescribing can contribute to population-level harm when long-term effectiveness is uncertain and risks are underestimated. Increased prescribing was followed by rising diversion, misuse, overdose deaths, and a wave of opioid use disorder. While the details differ between countries, the core lesson is consistent: opioids are powerful medicines that require strict indications, careful monitoring, and clear exit strategies.
Misconceptions About Opioids in Chronic Pain (and How to Reduce Harm)
Misconception 1: “If opioids don’t work, the dose just needs to be higher.” Dose escalation often increases harm more than benefit. A better approach is to reassess diagnosis, function, sleep, mood, and the wider pain plan.
Misconception 2: “All patients on opioids become addicted.” Addiction is not inevitable, but it is a real risk that increases with dose, duration, and vulnerability. Risk tools help but cannot perfectly predict outcomes.
Misconception 3: “Reducing opioids always worsens pain.” Many patients fear tapering. In practice, pain does not automatically worsen, and some patients improve as sedation reduces and opioid-induced hyperalgesia is addressed.
Mitigation strategies: Clear goals (function first), time-limited trials, lowest effective dose, frequent review, careful co-prescribing, and early planning for tapering if benefit is not meaningful.
Opioid Patches: Why They Are Commonly Misunderstood
Opioid patches are frequently misunderstood as being “mild” or “safer” than tablets because they deliver medication through the skin. In reality, patches deliver opioid continuously over days, can be difficult to titrate, and can still cause significant sedation, constipation, hormonal effects, and respiratory depression—especially when combined with other sedatives or used in opioid-naïve or frail patients.
It is also important to understand that many transdermal opioid systems were developed primarily for severe, persistent pain in settings such as cancer pain and palliative care, where stable background analgesia is required and the risk-benefit balance may differ from long-term non-cancer pain. Over time, patches migrated into wider use for persistent non-cancer pain, influenced by prescribing culture, limited alternatives, variable clinician familiarity with opioid potency, and in some settings the historic commercial promotion of opioid products.
Key message: Side effects depend on total opioid exposure and individual vulnerability—not the delivery method. A patch can represent a very high opioid load.
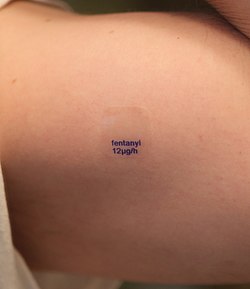
Fentanyl Patches: Extremely Potent—Often Under-Recognised
Fentanyl is an extremely potent opioid. A frequent safety problem is underestimating how strong fentanyl patches are and assuming that a “small patch” equals a “small dose.”
For example, a 25 micrograms/hour fentanyl patch is approximately equivalent to 60 mg of oral morphine in 24 hours. That is a substantial opioid dose delivered continuously.
Safety essentials: Fentanyl patches are not suitable for opioid-naïve patients. They take time to reach peak/steady-state levels, so dose changes should not be made too quickly. Heat (fever, hot baths, sauna, sunbeds, heat packs) can increase absorption and raise the risk of opioid toxicity, including respiratory depression.
Education gap: Incorrect prescribing and unsafe use often arise from lack of familiarity with opioid equivalence and insufficient patient education about potency, interactions, driving safety, and heat exposure.
Opioid Equianalgesic Doses (Educational Guide)
Important safety note: These conversions are approximate guides only. Inter-individual variation is significant, and switching opioids should be supervised by an experienced clinician. When switching between opioids, the calculated equi-analgesic dose should usually be reduced (often by ~25–50%) for safety, and by at least 50% in high-dose conversions or in frail/older patients. Methadone conversions require specialist advice.
| Opioid / Formulation | Approx. Equivalent to 10 mg Oral Morphine | Notes |
|---|---|---|
| Morphine (oral) | 10 mg | Reference standard. |
| Oxycodone (oral) | 6.6 mg | Approx. 1.5× morphine potency (guide). |
| Hydromorphone (oral) | 2 mg | High potency; caution when switching. |
| Codeine phosphate (oral) | 100 mg | Variable metabolism; effect can be unpredictable. |
| Dihydrocodeine (oral) | 100 mg | Approximate conversion. |
| Tramadol (oral) | 100 mg | Dual mechanism; conversion is approximate. |
| Tapentadol (oral) | 25 mg | Approximate conversion. |
Source: Faculty of Pain Medicine (Opioids Aware) dose equivalents and changing opioids guidance.
Fentanyl Patch Equivalence (Oral Morphine Equivalent, OME)
Key message: Fentanyl patches are extremely potent and should not be used in opioid-naïve patients. They can take time to reach peak/steady-state levels, so dose increases should not be made too quickly, and patients must be counselled carefully.
| Fentanyl Patch (micrograms/hour) | Approx. Oral Morphine (mg/day) |
|---|---|
| 12 | 30 |
| 25 | 60 |
| 50 | 120 |
| 75 | 180 |
| 100 | 240 |
Source: Faculty of Pain Medicine (Opioids Aware) transdermal fentanyl equivalence table.
Transdermal Buprenorphine (Weekly Patches): Approximate Morphine Equivalents
Buprenorphine patches are available in different strengths and schedules. The table below shows commonly used weekly strengths and approximate oral morphine equivalents.
| Buprenorphine Patch (micrograms/hour) | Approx. Oral Morphine (mg/day) |
|---|---|
| 5 | 12 |
| 10 | 24 |
| 20 | 48 |
Sources: Faculty of Pain Medicine (Opioids Aware) and West Midlands Palliative Care buprenorphine guidance.
Opioid Deprescribing: Why Reducing Opioids Does Not Necessarily Increase Pain
Deprescribing means supervised dose reduction when the harms of opioid therapy outweigh benefits, or when goals are not being met. A key message for patients is that tapering does not automatically mean worsening pain.
Some patients experience meaningful improvement during gradual reduction: clearer thinking, better energy, improved sleep, fewer gastrointestinal symptoms, improved hormonal symptoms, and less opioid-induced hyperalgesia. For others, pain may be similar but function improves as sedation and “fog” reduce.
Good practice is individualised and gradual, with shared decision-making, frequent review, pauses when needed, withdrawal management, and strong psychosocial support. Rapid, inflexible tapering can be harmful and is not the goal.
How Deprescribing Is Done Safely (A Practical Overview)
1) Agree the plan: Establish goals (function, sleep, activity), the pace of taper, and what will happen if symptoms flare.
2) Go slowly: Gradual reductions with the option to pause are usually safer and better tolerated than rapid tapering.
3) Monitor what matters: Pain interference, function, mood, sleep, and withdrawal symptoms—not just a pain score.
4) Reduce risk: Review sedating co-medications; consider naloxone education where appropriate; warn that tolerance falls during taper, so returning to old doses can increase overdose risk.
5) Strengthen the alternatives: Optimise non-opioid medications (when appropriate), rehabilitation, pacing, pain education, psychological strategies, and targeted treatments based on diagnosis.
When Opioids Might Still Be Appropriate
Opioids are not “never” medications. They can be appropriate in selected cases—especially short-term use for severe acute pain, cancer pain, and end-of-life care, and occasionally in carefully selected chronic pain situations where benefits are clear, goals are met, dose remains low, and risks are actively managed.
The key is that long-term opioid therapy should not drift into a default plan. It should be a deliberate, reviewed strategy with measurable benefit and a clear exit route if benefit is not sustained.
Pain Spa: Specialist Support for Long-Term Opioid Management and Tapering
At Pain Spa, Dr Krishna has extensive experience in reviewing and managing patients who have been on long-term opioids for persistent pain. This includes careful assessment of the original indication, current benefit versus harm, opioid burden (including patches), co-prescribed sedatives, and the wider drivers of persistent pain.
Where ongoing opioid therapy is not providing meaningful benefit—or where risk is high—Dr Krishna can support a structured, gradual opioid reduction plan, paced to the individual patient, with regular monitoring and practical strategies to minimise withdrawal and flare-ups. The aim is to improve safety, function, and quality of life, not to “force” dose reduction.
For many patients, combining medication review with targeted pain treatments and rehabilitation provides a safer and more effective long-term pathway than continuing dose escalation.
Key Take-Home Messages
• Long-term opioids for non-cancer pain usually offer limited sustained benefit and can cause cumulative harms.
• Risks increase with higher dose, longer duration, and sedating co-medications.
• Patches are not “mild by default”—and fentanyl patches in particular represent a very high opioid load.
• Deprescribing can be safe and beneficial when done gradually, collaboratively, and with appropriate support.
• The goal is better function and quality of life, using the right tools at the right time—not medication escalation as the only plan.