MCAS, Dysautonomia and Stellate Ganglion Block (SGB): Exploring the Neuro-Immune Link
Mast Cell Activation Syndrome (MCAS) and Stellate Ganglion Block: Understanding the Neuro-Immune Connection
Introduction
Mast Cell Activation Syndrome (MCAS) is an increasingly recognised condition in which mast cells release their chemical mediators in an exaggerated or inappropriate way. These mediators include histamine, tryptase, prostaglandins and leukotrienes, and when released in excess they can drive complex, fluctuating, multi-system symptoms that are often difficult to diagnose and manage. MCAS can affect the skin, gut, cardiovascular system, airways and nervous system, and in severe cases can produce anaphylaxis-like episodes.
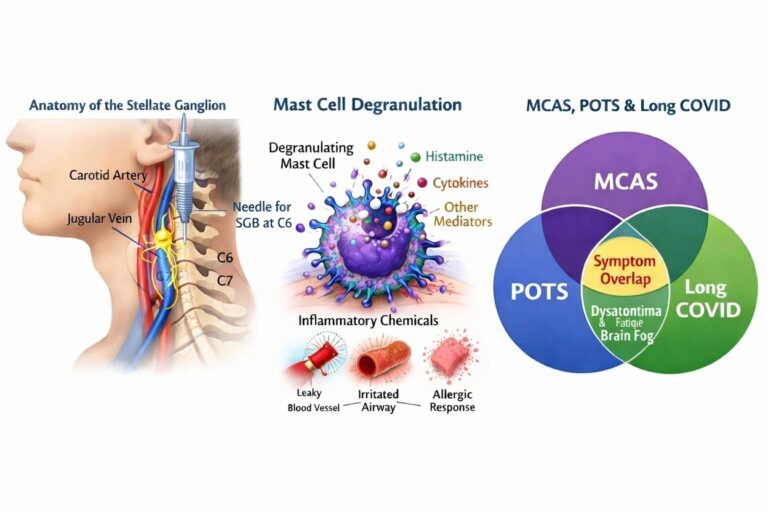
At Pain Spa, we see many patients with overlapping conditions such as MCAS, dysautonomia, POTS and Long COVID, where the nervous system and immune system appear tightly intertwined. This has led to growing interest in the neuro-immune dimension of chronic illness and in whether selected autonomic interventions may have a role in carefully chosen cases.
One such intervention is the stellate ganglion block (SGB). SGB is an established procedure in pain medicine and autonomic modulation, but it is important to state clearly at the outset that it is not an established treatment for MCAS. In MCAS, any use of SGB is off-label, meaning used outside its standard indications, and remains exploratory.
What Is Mast Cell Activation Syndrome (MCAS)?
Mast cells are part of the immune system and are found throughout the body, particularly in tissues that interface with the outside world such as the skin, airways, gut and blood vessels. Their normal role is protective: when they detect a perceived threat, they release mediators that help coordinate inflammation and immune defence.
In MCAS, mast cells appear to become over-sensitive or dysregulated, releasing mediators too easily or inappropriately, often without a clear trigger. This can lead to recurrent episodes of flushing, itching, swelling, diarrhoea, abdominal pain, tachycardia, wheeze, dizziness and sometimes severe allergic-type reactions affecting more than one body system at the same time.
It is helpful to distinguish MCAS from other mast-cell disorders. In mastocytosis, there is an abnormal increase in mast cell numbers. In hereditary alpha tryptasaemia syndrome (HATS), elevated tryptase is linked to inherited overproduction. In MCAS, mast cell numbers are usually normal, but their activation and mediator release are abnormal.
Common Symptoms and Clinical Features of MCAS
Because mast cells are present in many tissues, MCAS can produce a wide range of symptoms, often affecting several systems at once.
Dermatological symptoms may include flushing, itching, urticaria, skin sensitivity and angioedema.
Cardiovascular symptoms may include tachycardia, low blood pressure, light-headedness, presyncope or syncope.
Gastrointestinal symptoms may include abdominal pain, bloating, diarrhoea, constipation, nausea or vomiting.
Respiratory and ENT symptoms may include nasal congestion, wheeze, throat tightness and shortness of breath.
Neurological and systemic symptoms may include fatigue, headaches, “brain fog”, temperature instability and feeling generally unwell.
In more severe cases, MCAS can present with anaphylaxis-like episodes, with sudden multi-system involvement and the need for urgent medical treatment. MCAS is generally considered when symptoms involve two or more body systems together.
Why MCAS Is Difficult to Diagnose
MCAS is difficult to diagnose because symptoms vary widely between patients and may fluctuate over time in the same person. Its clinical picture also overlaps with other conditions such as POTS, dysautonomia, allergic disorders, gastrointestinal syndromes and Long COVID.
Current diagnostic approaches usually rely on three broad elements: typical recurrent symptoms suggestive of mast cell activation, objective evidence of mediator release on testing, and improvement with mast-cell directed treatment.
Serum tryptase is often discussed, but interpretation requires care. Tryptase usually needs to be measured more than once, including during or shortly after a flare, and raised levels are not specific to MCAS. Equally important, some patients may have symptoms strongly suggestive of mast cell activation even when routine tests are normal between flares, so a normal result does not automatically exclude the condition. Elevated histamine alone is also not sufficient to diagnose MCAS.
For this reason, diagnosis usually depends on careful history-taking, consideration of differentials, assessment by an appropriate specialist, and sometimes a therapeutic trial of treatment.
Current Evidence-Based Management of MCAS
Management of MCAS usually combines trigger reduction, medication and practical lifestyle strategies. There is no single cure, and treatment often needs to be individualised.
Commonly used treatments include H1 antihistamines such as cetirizine, loratadine and fexofenadine, H2 blockers such as famotidine, mast-cell stabilisers such as cromolyn sodium, leukotriene-directed therapies such as montelukast in selected cases, and biologics such as omalizumab in selected specialist settings. Patients at risk of severe reactions may also require epinephrine. These treatments should be used under the supervision of the appropriate prescribing clinician.
Trigger identification remains extremely important. Common triggers may include foods, alcohol, heat, infection, exercise, physical pressure, emotional stress, certain medications, environmental chemicals, perfumes and hormonal shifts, although trigger profiles vary markedly from one patient to another. A symptom and trigger diary can be very useful. Some patients also explore low-histamine or other specialist diets under clinical supervision.
One practical challenge is that some medicines themselves, or their additives and excipients, may trigger symptoms in sensitive patients. This means that successful treatment may take time and often involves careful, supervised trial and adjustment rather than a single standard prescription.
The Autonomic Nervous System and Mast Cell Activation
The autonomic nervous system regulates heart rate, blood pressure, gut motility, sweating, temperature control and many unconscious body functions. It has two main branches: the sympathetic nervous system, often described as “fight or flight”, and the parasympathetic system, often described as “rest and digest”.
Mast cells sit close to nerves and blood vessels and can both respond to, and influence, neural signalling. Experimental work has shown that neuropeptides and other nerve-derived signals can influence mast-cell behaviour, helping explain why stress and autonomic arousal may worsen symptoms in some patients.
Clinically, many patients with MCAS describe symptom worsening during stress, illness, orthostatic strain or other situations associated with autonomic arousal. This has contributed to growing interest in the relationship between MCAS, dysautonomia, POTS and post-viral syndromes such as Long COVID. The exact causal relationship is not fully understood, but the overlap is striking enough that autonomic imbalance is now part of the clinical conversation in many complex multi-system patients.
The Stellate Ganglion: A Key Autonomic Control Centre
The stellate ganglion is part of the cervical sympathetic chain in the lower neck. It plays a key role in sympathetic signalling to the head, neck, upper chest and upper limbs, and through wider autonomic pathways can influence cardiovascular responses, vascular tone and stress-related physiology.
More recent literature has broadened how the stellate ganglion is viewed. Rather than being considered only as a relay for sympathetic output, it is increasingly discussed in the context of neuroimmune regulation, with evidence that abnormal stellate ganglion activity and inflammation may interact in disease states.
Stellate Ganglion Block (SGB): What the Procedure Does?
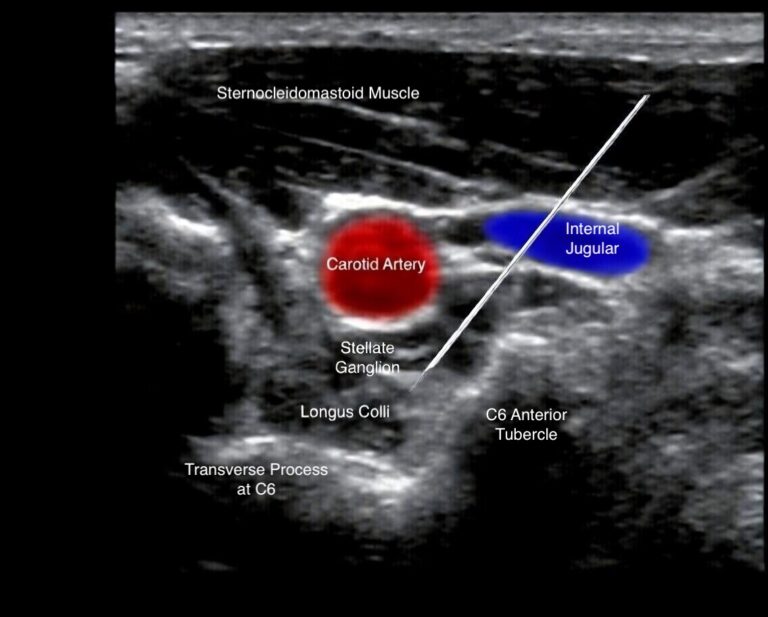
A stellate ganglion block is a minimally invasive injection of local anaesthetic around the stellate ganglion to temporarily reduce sympathetic activity in that region. In modern practice, it is usually performed under real-time ultrasound guidance to improve accuracy and safety.
During the procedure, a fine needle is guided to the target area in the neck and a small volume of local anaesthetic is injected. Patients are monitored during and after the procedure, and most go home the same day once stable.
The aim is not simply to numb tissue, but to dampen sympathetic outflow and alter autonomic balance. In pain medicine, SGB has long been used for sympathetically maintained pain and certain vascular or autonomic conditions. In MCAS, however, any use of SGB is off-label and experimental.
Neuro-Immune Modulation: How SGB May Influence Inflammation?
The main reason SGB has attracted interest in inflammatory and autonomic disorders is biological plausibility. Sympathetic nerves innervate primary and secondary immune organs, including bone marrow, thymus, spleen and lymphoid tissue. Immune cells express adrenergic receptors and can respond to autonomic signals.
A 2020 hypothesis paper proposed that by interrupting sympathetic input to immune organs, SGB may influence immune signalling more broadly. The authors discussed possible reductions in pro-inflammatory cytokines such as IL-1β, IL-6 and TNF-α, increases in anti-inflammatory signalling such as IL-10 and TGF-β, and shifts in immune regulation via central reflex-like pathways involving the insula, amygdala, hippocampus and locus ceruleus.
This does not prove that SGB treats MCAS, nor does it directly show suppression of mast-cell disease in patients with MCAS. What it does provide is a plausible neuro-immune framework for why sympathetic modulation might influence some inflammatory or autonomic symptom clusters in selected overlap cases.
Evidence That Stellate Ganglion Activity Influences Inflammation
A 2024 review on the stellate ganglion, inflammation and arrhythmias reinforces the broader concept that stellate ganglion activity and inflammatory pathways can interact. The review describes the stellate ganglion as an important neuroimmune structure and discusses evidence that abnormal stellate ganglion activity may contribute to sympathetic overactivation in inflammatory states. It also reviews preclinical and clinical observations suggesting that interventions targeting stellate ganglion activity may influence inflammatory signalling.
Some of the reviewed evidence comes from cardiovascular and neuroinflammatory models rather than MCAS. That matters. These papers support the principle of neuroimmune interaction, but they do not demonstrate efficacy for MCAS. In other words, the rationale is scientifically interesting, but the disease-specific evidence gap remains.
Evidence for Stellate Ganglion Block in Related Conditions
Long COVID
This is currently the strongest related-condition evidence relevant to the MCAS discussion. A 2025 prospective pilot study described ten adults with Long COVID and ME/CFS who underwent weekly stellate ganglion blocks for three weeks. Over the following two months, improvements were reported in cognition, orthostatic intolerance, physical and social functioning, vitality and POTS-related symptoms. Not all symptoms improved, and the study was small, but it provided encouraging early signals in post-viral dysautonomia.
Earlier work from the same clinical group had also reported symptom improvements in Long COVID, including fatigue, dizziness on standing, mood symptoms, post-exertional malaise, focus, memory, smell and taste, with some benefits reported to persist for weeks or months.
Dysautonomia and POTS
Formal data remain limited, but the rationale for using SGB in severe dysautonomia and POTS-like pictures is based on reducing sympathetic overactivity and attempting to “reset” autonomic balance. This is especially relevant in patients whose main burden of illness comes from orthostatic intolerance, tachycardia, hyperarousal and post-viral autonomic dysfunction. The evidence here remains emerging rather than definitive.
PTSD and Other Autonomic Conditions
SGB has also been explored in PTSD, where autonomic hyperarousal is a major feature, and it has longstanding use in other pain and autonomic contexts. This is relevant because it supports the broader concept of autonomic modulation, even though PTSD is obviously very different from MCAS.
Could Stellate Ganglion Block Help Patients with MCAS?
From a mechanism-based perspective, there is a plausible link. If sympathetic overactivity contributes to mast cell triggering, and if some patients with MCAS have major overlap with POTS, dysautonomia or Long COVID, then reducing sympathetic drive might indirectly reduce some of the inputs that aggravate symptoms.
That said, this remains a hypothesis. There are currently no randomised controlled trials, no prospective cohorts focused on primary MCAS, and no published MCAS-specific case series demonstrating that SGB reliably improves core MCAS symptoms such as flushing, gastrointestinal reactivity, mediator-driven episodes or anaphylactoid events. The idea is biologically plausible, but not yet proven.
For this reason, SGB should not be presented as a standard or established treatment for MCAS.
What the Current Scientific Evidence Actually Shows
At present, the evidence supports the following balanced position:
SGB has established roles in some pain and sympathetic conditions.
It has emerging evidence in Long COVID, ME/CFS overlap, dysautonomia-related syndromes and PTSD.
It has a plausible neuro-immune rationale based on sympathetic modulation and inflammatory signalling.
It does not yet have direct clinical evidence proving benefit in MCAS.
As of 2026, any use of SGB for MCAS should therefore be viewed as an experimental neuro-immune modulation strategy, not a validated mast-cell treatment.
When Might SGB Be Considered in Patients with MCAS?
SGB is not first-line treatment for MCAS and will not be appropriate for most patients. However, there may be rare scenarios where considering it as an adjunct is reasonable.
These might include:
MCAS with severe dysautonomia, where orthostatic intolerance, tachycardia and autonomic hyperarousal are major drivers of disability.
MCAS overlapping with Long COVID, particularly where dysautonomia is prominent and standard measures have plateaued.
MCAS with established POTS or related autonomic dysfunction, where sympathetic overactivity appears central to symptom flares.
Patients with refractory symptoms despite optimised standard MCAS treatment, who have been assessed by appropriate clinicians and understand the uncertainties.
At Pain Spa, this would never replace standard medical care. For example, in a patient with MCAS-like symptoms and marked POTS or Long COVID features, we would strongly recommend first to optimise standard treatment, trigger management and non-invasive autonomic strategies before considering procedures such as SGB.
Safety and Risks of Stellate Ganglion Block
When performed by experienced clinicians under real-time ultrasound guidance, stellate ganglion block (SGB) is generally considered a safe procedure.
Most side effects are temporary and expected, and occur because the sympathetic nerves in the neck are temporarily blocked. These effects typically resolve as the local anaesthetic wears off.
Common temporary effects may include:
Horner syndrome on the treated side (a drooping eyelid and smaller pupil)
Warmth or flushing of the face
Mild hoarseness
A temporary sensation of difficulty swallowing or a “lump in the throat”
These effects are usually short-lived and settle within a few hours.
When SGB is performed using ultrasound guidance by experienced clinicians, serious complications are very rare. Careful technique, appropriate patient selection, and monitoring during and after the procedure help ensure that the treatment is carried out as safely as possible.
Key Take-Home Messages
MCAS is a multi-system condition driven by inappropriate mast-cell activation, producing fluctuating symptoms that can be difficult to diagnose and manage. Standard care focuses on trigger avoidance, antihistamines, mast-cell stabilisers, leukotriene-directed therapies, selected biologics and emergency planning where needed.
The autonomic nervous system and mast cells appear to be closely linked, and dysautonomia is commonly seen in overlap states such as MCAS, POTS and Long COVID. SGB is an established intervention for some pain and autonomic conditions and has shown promising early signals in related disorders such as Long COVID and PTSD, but it has not been formally studied in MCAS.
Any consideration of SGB in MCAS should therefore be cautious, individualised and part of a broader evidence-based treatment plan.
The Pain Spa Approach to Complex Neuro-Immune Disorders
At Pain Spa, our approach to complex syndromes such as MCAS, Long COVID and dysautonomia is mechanism-based and collaborative. We aim to identify autonomic drivers, immune contributors, pain generators and functional barriers, and then build an integrated plan rather than relying on a single intervention.
Where appropriate, we may consider interventional procedures such as ultrasound-guided stellate ganglion block alongside medical treatment, lifestyle measures and input from other specialists. Any decision to proceed is made through shared decision-making after a detailed discussion of the potential benefits, risks and uncertainties.
Dr Krishna’s approach is to use precise ultrasound-guided technique, careful patient selection and long-term follow-up, particularly in patients with overlapping autonomic and pain presentations.
Frequently Asked Questions
What triggers mast cell activation syndrome?
Triggers vary, but may include foods, alcohol, heat, infections, medications, hormonal shifts, exercise, physical pressure, environmental chemicals and psychological stress. Some patients identify clear triggers, while others seem to react unpredictably.
Can stress worsen MCAS symptoms?
Yes. Stress activates the sympathetic nervous system and is commonly reported as a trigger for flares. In susceptible individuals, autonomic arousal may worsen flushing, tachycardia, gut symptoms and general reactivity.
Is stellate ganglion block an established treatment for MCAS?
No. There are currently no clinical trials or dedicated case series specifically evaluating SGB in MCAS patients. Its use in this context is off-label and experimental.
How long does a stellate ganglion block last?
The local anaesthetic effect lasts hours, but in some conditions the clinical benefit may outlast the anaesthetic, sometimes for days, weeks or longer. Duration is variable and cannot be guaranteed.
Who might benefit from SGB?
Potential candidates usually include patients with severe autonomic dysfunction, POTS, Long COVID overlap or related sympathetic hyperarousal states. In rare cases, selected MCAS patients with major dysautonomia overlap may be considered after specialist assessment and optimisation of standard care.
Is SGB safe?
When performed by experienced clinicians using ultrasound guidance, SGB is generally safe, but complications can occur. Temporary side effects are more common than serious ones, which are rare but important.
Future Research Directions
There is a clear need for well-designed studies exploring SGB and other autonomic modulation techniques in patients with MCAS and overlapping dysautonomia. Future research should include better phenotyping, robust outcome measures, longer follow-up and ideally biomarker work to identify which symptom patterns are most likely to respond.
More broadly, ongoing neuro-immune research is beginning to clarify how nerves and immune cells communicate in chronic inflammatory and post-viral conditions. That line of work is highly relevant to Pain Spa’s interest in mechanism-based care for complex chronic illness.
References
The following references include consensus papers on MCAS, educational clinical overviews, and key literature on stellate ganglion block, neuro-immune modulation and Long COVID-related dysautonomia.
1. Weiler CR, Austen KF, Akin C, et al. Mast cell activation syndrome: tools for diagnosis and differential diagnosis. Journal of Allergy and Clinical Immunology: In Practice. 2019.
2. Afrin LB, Butterfield JH, Raithel M, Molderings GJ. Often seen, rarely recognised: mast cell activation disease — a guide to diagnosis and therapeutic options. Annals of Medicine. 2016.
3. Cleveland Clinic. Mast Cell Activation Syndrome (MCAS): Symptoms, Causes and Treatment. Updated 2024.
4. Lipov EG, Gluncic V, Lukic IK, Candido KD. How does stellate ganglion block alleviate immunologically-linked disorders? Medical Hypotheses. 2020;139:110000.
5. Lei Q, Jiang Z, Shao Y, Liu X, Li X. Stellate ganglion, inflammation, and arrhythmias: a new perspective on neuroimmune regulation. Frontiers in Cardiovascular Medicine. 2024;11:1453127.
6. Liu J, Duricka DL. Stellate ganglion block reduces symptoms of Long COVID: a case series. 2022.
7. Duricka DL, Liu J, et al. Stellate ganglion block reduces symptoms of SARS-CoV-2-induced ME/CFS: a prospective cohort pilot study. 2025.
8. Mulvaney SW, Lynch JH, Hickey MJ, et al. Stellate ganglion block used to treat symptoms associated with post-traumatic stress disorder: a review of published evidence. Military Medicine. 2014.
9. Theoharides TC, Tsilioni I, Ren H. Recent advances in our understanding of mast cell activation — or should it be mast cell mediator disorders? Expert Review of Clinical Immunology. 2019.
If you suffer from MCAS or dysautonomia and would like an assessment for SGB injections, please contact Pain Spa at clinic@painspa.co.uk or visit www.painspa.co.uk for further information.