Low-Dose Naltrexone (LDN) in Chronic Pain, Long COVID, ME/CFS, Fibromyalgia and PTSD

Low-Dose Naltrexone (LDN): Mechanisms, Evidence and Practical Clinical Use in Chronic Pain, Long COVID, ME/CFS, Fibromyalgia and PTSD
Low-dose naltrexone (LDN) has emerged as an increasingly important off-label option in chronic pain and related neuroimmune conditions. This article explains how it works, where the evidence is strongest, how it may fit into clinical practice, how it should be dosed, and why opioid compatibility and perioperative planning matter.
Low-Dose Naltrexone (LDN): Why Is It Getting Attention in Chronic Illness and Chronic Pain?
Low-dose naltrexone has become one of the most discussed off-label treatments in chronic pain and complex multisystem conditions over recent years. The reason for this growing interest is not that it is new in an absolute sense, but that our understanding of chronic pain has evolved. Increasingly, conditions such as fibromyalgia, chronic widespread pain, long COVID, chronic fatigue syndrome / myalgic encephalomyelitis (ME/CFS), and some trauma-related syndromes are being understood not simply as problems of damaged tissues, but as conditions involving central sensitisation, altered pain processing, neuroinflammation, autonomic dysregulation, and immune disturbance.
In this context, LDN is appealing because it does not fit neatly into the usual categories of pain medication. It is not a conventional analgesic, not an anti-inflammatory in the usual NSAID sense, and not an opioid. Instead, it appears to act as a neuroimmune modulator. That has led to interest in conditions where patients often have pain, fatigue, brain fog, poor sleep, hypersensitivity, post-exertional flares, and a poor response to standard drug therapy.
At the same time, it is important to be honest about where the field currently stands. LDN is promising, but it is not a miracle drug and it is not yet supported by large, definitive trials across all of these indications. The evidence is strongest in fibromyalgia and chronic pain-related literature, and more preliminary in long COVID and ME/CFS. For PTSD, the rationale is more indirect and theoretical than firmly evidence-based. That balanced framing is essential if we are to discuss LDN responsibly.
What Is LDN and How Is It Different from Standard-Dose Naltrexone?
Naltrexone is classically known as an opioid receptor antagonist used at standard doses, typically 50 mg or more, for alcohol and opioid dependence. At those doses it produces sustained opioid receptor blockade. Low-dose naltrexone, by contrast, usually refers to doses in the 0.5 mg to 4.5 mg range, although real-world prescribing sometimes extends below or above this depending on the clinical situation. In that lower range, naltrexone appears to behave very differently pharmacodynamically.
This distinction matters. LDN should not be thought of simply as “less naltrexone.” At low doses, its action is not just a scaled-down version of full-dose opioid blockade. Instead, it appears to produce a brief, partial opioid receptor blockade and, importantly, may also affect non-opioid pathways such as glial cell activity and Toll-like receptor 4 signalling. That is why LDN has become relevant in chronic pain and immune-related disorders, whereas standard-dose naltrexone is used for addiction medicine.
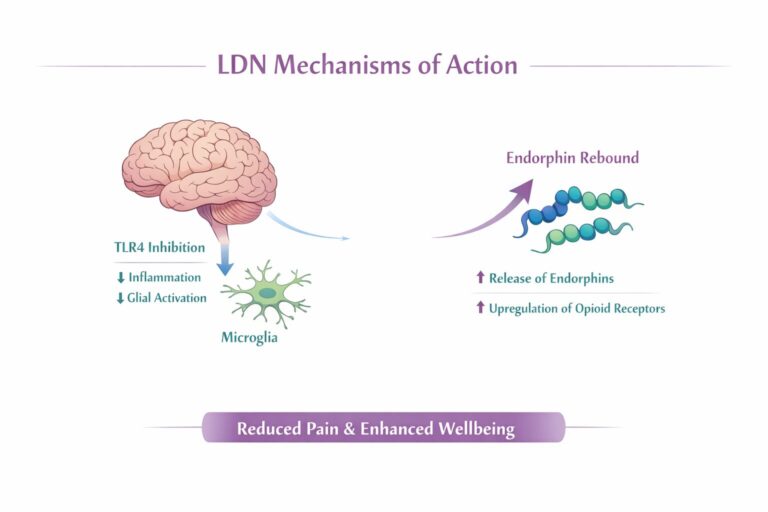
How LDN Works: Mechanism of Action
At a Glance: How LDN May Work
Temporary opioid receptor blockade → endorphin rebound → microglial modulation → reduced neuroinflammation → less central sensitisation → possible improvement in pain, fatigue, sleep and brain fog.
Transient opioid receptor blockade and the “rebound” endorphin effect
One of the long-standing theories behind LDN is that a short period of opioid receptor blockade leads to a compensatory rebound in endogenous opioid activity. Put simply, the body senses a temporary reduction in opioid signalling and responds by increasing endorphin production and possibly receptor sensitivity. That rebound may contribute to improvements in pain modulation, wellbeing, and in some patients sleep and mood.
This mechanism is attractive because it helps explain why a medication known as an opioid antagonist can, paradoxically, help some chronic pain patients. The key point is that the blockade is brief rather than continuous, and that the downstream biological response may be more important than the blockade itself. However, this is only one part of the story and does not adequately explain the full range of observed clinical effects.
Microglial modulation and neuroinflammation
The most important modern explanation for LDN’s use in chronic pain is probably its effect on microglia. Microglia are the resident immune cells of the central nervous system. When activated, they can release pro-inflammatory cytokines, excitatory mediators and other substances that amplify pain, worsen fatigue, disrupt sleep, and contribute to cognitive symptoms such as brain fog. Chronic microglial activation has therefore become a major theme in conditions such as fibromyalgia, nociplastic pain, ME/CFS and long COVID (Younger et al., 2014).
LDN has been described as a glial modulator, and one of its key proposed actions is suppression of this maladaptive microglial activation. If that is correct, the drug is not simply reducing pain signals in the short term; it is altering the inflammatory environment in which central sensitisation is being maintained. That is precisely why it is being discussed in syndromes characterised by pain, fatigue, hypersensitivity, and cognitive dysfunction rather than just classical inflammatory arthritis or acute injury.
Toll-like receptor 4 (TLR4) and immune signalling
Another central mechanism is antagonism of Toll-like receptor 4, or TLR4. TLR4 is involved in innate immune signalling and has been implicated in neuroinflammatory cascades. The chronic pain review by Younger and colleagues specifically discusses naltrexone’s antagonistic action at non-opioid receptors such as TLR4 on microglia, linking this to reduced inflammatory and excitatory signalling (Younger et al., 2014).
This matters clinically because it fits the symptom clusters we see in real patients. Pain, fatigue, cognitive dysfunction, malaise, poor sleep and sensory hypersensitivity often travel together. If LDN does reduce TLR4-driven inflammatory signalling, that offers a biologically plausible explanation for why some patients report improvements across several domains rather than pain alone.
Why LDN may help nociplastic and centrally sensitised pain
Many patients seen in pain clinics do not have pain that is fully explained by ongoing tissue damage. Instead, they have nociplastic pain: altered nociceptive processing in the nervous system, often accompanied by hyperalgesia, fatigue, poor restorative sleep, stress intolerance, and multiple overlapping symptoms. Fibromyalgia is the classic example, but the same framework increasingly applies to chronic widespread pain, hypermobility-related pain, post-viral pain states, and some pelvic pain and trauma-related syndromes.
LDN fits this space well because it may modify the biology of sensitisation rather than simply trying to “block pain.” That makes it particularly attractive when standard analgesics, especially opioids, have either failed or risk making the overall clinical picture worse.
Ultra-low-dose, very-low-dose and low-dose naltrexone: what is the difference?
This is an area where confusion is common. “Low-dose naltrexone” generally refers to roughly 1 to 5 mg daily, though many clinicians practically use 0.5 to 4.5 mg as a common working range. “Very-low-dose naltrexone” sits below that, roughly in the microgram to under-1 mg range, while “ultra-low-dose naltrexone” usually refers to extremely small doses, often used experimentally alongside opioids rather than as stand-alone therapy. These dose ranges likely engage different mechanisms and should not be treated as interchangeable.
That distinction becomes especially important when discussing opioids. Standard LDN and ultra-low-dose naltrexone are not the same therapeutic strategy. The former is generally avoided with regular opioids; the latter has been explored experimentally to modify opioid signalling. These broader pharmacological themes are also summarised in review literature on LDN (Toljan & Vrooman, 2018).
Why LDN Is Being Considered in Conditions with Pain, Fatigue and Neuroimmune Dysregulation
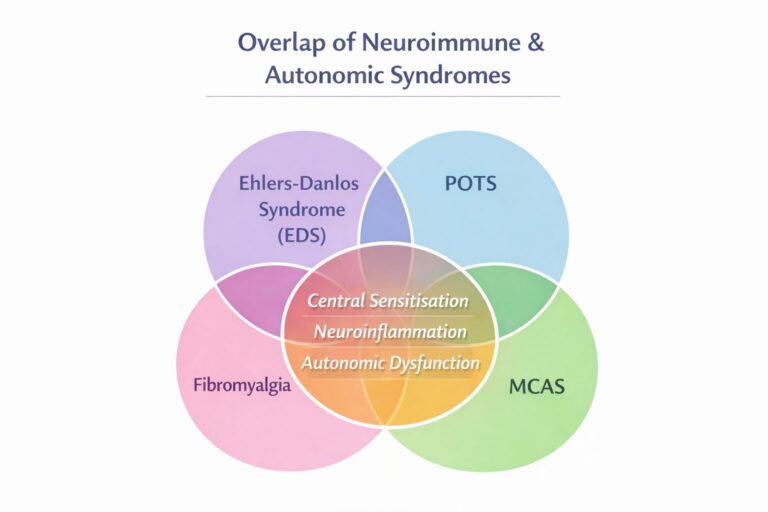
The growing list of conditions in which LDN is being discussed may seem odd at first glance. Fibromyalgia, Crohn’s disease, long COVID, ME/CFS, complex regional pain syndrome, multiple sclerosis, hypermobility-related pain and mast cell-related syndromes do not look identical on paper. Yet they all involve, to varying degrees, neuroimmune signalling, altered autonomic regulation, inflammation-related symptom amplification, and reduced resilience of the nervous system.
That overlap is particularly obvious in clinic. A patient with long COVID may also meet criteria for ME/CFS. A patient with fibromyalgia may have dysautonomia. A patient with hypermobile EDS may have chronic pain, MCAS-type symptoms, bowel hypersensitivity and orthostatic intolerance. In that setting, LDN is attractive not because it is diagnosis-specific, but because it may target a shared network of central sensitisation and neuroimmune dysregulation.
Overlap Syndromes Snapshot
Fibromyalgia, Long COVID, ME/CFS, EDS, POTS and MCAS often overlap through shared mechanisms such as central sensitisation, autonomic dysfunction, neuroinflammation and multisystem symptom amplification.
Overlap Syndromes: Ehlers-Danlos Syndrome (EDS), POTS and Mast Cell Activation Syndrome (MCAS)
This is one of the most clinically useful areas to understand, because these conditions often overlap and create exactly the kind of patient in whom LDN is considered. Patients with hypermobile EDS frequently present with chronic widespread pain, recurrent injury, central sensitisation, autonomic symptoms and gastrointestinal disturbance. POTS adds orthostatic intolerance, palpitations, fatigue and brain fog. MCAS brings flushing, reactivity, bowel symptoms, headaches, rashes, chemical sensitivity and major medication sensitivity. The result is often a patient who looks “complex” on paper but in reality has a recognisable pattern of multisystem dysregulation.
The LDN Research Trust discussion on EDS, POTS and MCAS is very useful not because it proves efficacy with high-level trials, but because it reflects the practical prescribing wisdom that has emerged from treating these overlap syndromes. The central message is simple: start very low, increase very slowly, and pay close attention to formulation. The page specifically notes that in these patients clinicians often begin with very small once-daily doses and increase perhaps every 14 days, often using liquid formulations to allow fine dose adjustment. It also highlights that excipients, flavourings and sweeteners can matter because some highly reactive patients worsen not from the active drug itself but from the formulation.
LDN in EDS
In EDS, LDN is not “fixing connective tissue.” It is much more likely helping the central pain and inflammatory amplification that often develops around chronic instability and recurrent mechanical stress. That is important because many hypermobile patients have both peripheral pain generators and a secondary central sensitisation picture. If LDN helps, it is most likely reducing the background amplification, improving flare threshold, and sometimes helping sleep or recovery rather than correcting the underlying hypermobility itself. The 2024 observational dosing study explicitly included chronic pain populations relevant to hypermobile EDS and reinforces how individualized dose finding can be in this group (Marcus et al., 2024).
LDN in POTS
POTS is more complex because its symptoms are not purely pain-based. Fatigue, orthostatic intolerance, tachycardia, brain fog and post-exertional worsening may dominate. LDN is not a substitute for fluid, salt, compression, pacing or formal dysautonomia management. However, where POTS overlaps with post-viral illness, pain, inflammatory symptoms or central sensitisation, LDN may have a role as part of a broader neuroimmune strategy. The long COVID literature is especially relevant here, because post-COVID cohorts clearly include patients with orthostatic and POTS-like symptoms alongside fatigue and brain fog.
LDN in MCAS
MCAS is where practical prescribing becomes particularly important. These patients are often the most medication-sensitive in the clinic. A perfectly reasonable standard starting dose for another patient may feel overwhelming to someone with MCAS. That is why the low-and-slow approach is so important. For some, 0.1 mg or 0.25 mg may be more appropriate than 1 mg or 1.5 mg. Liquids can be helpful, but the base and additives matter. Improvement, if it occurs, may include reduced overall inflammatory reactivity, fewer pain flares, better tolerance of daily activity, or less severe multisystem “crashes.” But expectations should be careful and titration respectful.
Practical message for overlap syndromes
In EDS/POTS/MCAS overlap, the goal is not aggressive dose escalation. The goal is to find the lowest useful dose that improves global system stability. That is where LDN can be surprisingly valuable: not as a blunt analgesic, but as a gentle central modulator in a patient group that often reacts badly to more conventional medications.
LDN in Chronic Pain
Chronic pain, central sensitisation and nociplastic pain
Chronic pain is increasingly understood as more than persistent nociceptive input. Many chronic pain patients develop amplification of the pain system itself. They become more pain-sensitive, more flare-prone, and more likely to experience fatigue, poor sleep and cognitive symptoms. This is one reason why conventional structural explanations often feel incomplete and why escalating analgesics can fail.
LDN is relevant here because it appears to address this amplified, sensitised biology. That is especially important in patients whose pain has become widespread, symmetrical, or accompanied by multiple central symptoms.
What the current evidence shows
The evidence in chronic pain is encouraging but not definitive. Reviews and small clinical studies suggest benefit in certain centralised pain states, particularly fibromyalgia and some inflammatory or neuroinflammatory conditions. The key message is that LDN is promising, inexpensive, and generally well tolerated, but still off-label and not yet validated by large, robust trials across the board.
Why response to dose can be highly individual
One of the most useful recent observations is that LDN does not behave like a one-size-fits-all medication. In the 2024 observational study of chronic musculoskeletal pain, the maximally effective dose varied widely, ranging from 0.1 mg to 6 mg per day, and the authors concluded that the effective dose is idiosyncratic and should be found by titration rather than assumption (Marcus et al., 2024). That is clinically very important. It means that failure at one fixed dose should not automatically be interpreted as failure of the treatment itself.
LDN in Fibromyalgia
Why fibromyalgia may respond to LDN
Fibromyalgia is the condition most closely associated with LDN in the chronic pain literature. That makes sense because fibromyalgia combines widespread pain, fatigue, poor sleep, cognitive dysfunction, dysautonomia and central hypersensitivity. It is one of the clearest clinical expressions of nociplastic pain and altered neuroimmune signalling.
Clinical trial and review evidence in fibromyalgia
The evidence base remains small but is among the strongest for any LDN indication in pain medicine. Younger and colleagues reported symptom improvement in early fibromyalgia studies, and later reviews have concluded that LDN may be effective and safe, while also highlighting that the studies are small and underpowered. The Australian systematic review notes that only a handful of prospective controlled studies exist, but the overall signal is sufficiently positive to justify clinical interest (Aitcheson et al., 2023).
What symptoms may improve: pain, fatigue, sleep and mood
Fibromyalgia patients who respond often report improvement across several domains rather than pain alone. That includes pain severity, fatigue, sleep quality, mental clarity and sometimes mood. This broader symptom response fits the underlying biology: if the drug is modulating central inflammatory signalling and endogenous opioid function, a multi-domain improvement is exactly what one might expect.
LDN in Long COVID
Why LDN has been proposed for long COVID
Long COVID has brought renewed attention to LDN because it is a syndrome in which patients frequently develop persistent fatigue, brain fog, pain, dysautonomia, sleep disruption and post-exertional worsening after infection. These symptoms strongly overlap with ME/CFS and fibromyalgia, suggesting shared mechanisms.
Fatigue, pain, brain fog and post-exertional symptom burden
This is where LDN becomes clinically interesting. Long COVID is not just “tiredness after infection.” It often includes persistent neuroimmune activation, orthostatic symptoms, sensory hypersensitivity and cognitive dysfunction. If LDN reduces microglial activation and inflammatory signalling, it becomes biologically plausible in exactly this symptom cluster.
What the early cohort and pilot studies show
The long COVID data are still early, but they are encouraging. In the 2024 pilot study combining LDN 4.5 mg/day with NAD+ in patients with persistent post-COVID fatigue, quality-of-life scores improved significantly over 12 weeks, fatigue scores improved, and 52% of patients were classified as responders. It was an open-label pilot and therefore far from definitive, but it supports the idea that LDN may help at least a subset of patients (Isman et al., 2024).
Overlap between Long COVID, fibromyalgia and ME/CFS
The overlap is now impossible to ignore. Long COVID, ME/CFS and fibromyalgia share pain, fatigue, post-exertional worsening, poor sleep, cognitive dysfunction and autonomic symptoms. That does not mean they are identical, but it does mean a treatment that appears useful in one may reasonably be explored in the others.
LDN in ME/CFS (Chronic Fatigue Syndrome / Myalgic Encephalomyelitis)
Why LDN is being explored in ME/CFS
ME/CFS is characterised by profound fatigue, post-exertional malaise, pain, autonomic dysfunction and cognitive symptoms. There are no licensed curative treatments, and standard drug approaches are often disappointing. That has led to considerable off-label use of LDN in this population.
Neuroimmune, inflammatory and autonomic hypotheses
The neuroimmune model of ME/CFS makes LDN a logical candidate. Even if the exact mechanism remains unsettled, the combination of immune disturbance, inflammatory signalling, autonomic dysfunction and central sensitisation aligns closely with LDN’s proposed biology.
Current evidence: case series, observational experience and limitations
The evidence remains limited. The BMJ case report series described varied responses, with doses ranging from 4 mg to 12 mg and outcomes ranging from partial symptom relief to major life-changing improvement. The Finnish retrospective experience cited in the long COVID paper also suggests a substantial proportion of ME/CFS patients report benefit, but these are not randomised controlled trials. So the evidence is promising, but still preliminary (Bolton et al., 2020).
Which symptoms may improve, and where uncertainty remains
Symptoms that may improve include fatigue, pain, sleep and cognitive clarity. However, the response is variable and post-exertional malaise remains a key issue. LDN should therefore be presented as a trial treatment rather than a guaranteed solution.
LDN in PTSD
Why LDN has been discussed in trauma-related conditions
The evidence for PTSD is much thinner than for fibromyalgia or chronic pain. Nonetheless, there is growing interest because trauma-related disorders often involve hyperarousal, sleep disruption, central sensitisation, autonomic dysregulation and overlapping chronic pain. These features create a theoretical rationale for LDN. The key word here is theoretical. The direct evidence base is currently limited. That should be stated clearly.
Neuroinflammation, sleep, hyperarousal and central sensitisation
If neuroinflammation contributes to hypervigilance, poor sleep and stress amplification, LDN may have a role in selected patients, particularly those with overlapping pain, fatigue or centrally sensitised symptoms. But at present this is more extrapolation than established evidence.
What is theoretical, what is indirect, and what is actually supported by evidence
The strongest honest statement is this: PTSD is not currently one of the best-evidenced indications for LDN. It may be considered in selected complex patients, especially where trauma, pain and central sensitisation overlap, but this should be framed cautiously and not presented with the same confidence as fibromyalgia or chronic pain.
What Dose of LDN Is Used?
Dose is one of the most important issues in LDN prescribing, and also one of the most misunderstood. The older literature often centres on 4.5 mg nightly because that was the most common study dose. But newer clinical experience and observational work make it clear that this is not a universal optimum. Some patients respond well below that, while others appear to need higher doses.
Typical LDN range
A practical clinical range is often 0.5 mg to 4.5 mg daily, though some papers describe 1 to 5 mg as the core LDN range.
Ultra-low-dose naltrexone
Ultra-low-dose naltrexone refers to microgram-level dosing and is generally discussed in relation to opioid co-therapy, not standard stand-alone LDN prescribing.
Very-low-dose titration for sensitive patients
Very-low-dose approaches below 0.5 mg are especially useful in sensitive patients, including some with ME/CFS, MCAS and significant autonomic instability. This is also where liquid preparations can be helpful.
When some clinicians use doses above 4.5 mg and up to 9–10 mg
Some patients, especially in ME/CFS case reports and real-world practice, appear to benefit from doses above 4.5 mg, occasionally much higher. That does not mean higher is better in general, only that the response curve is not fixed.
Why there may not be one “correct” dose for everyone
The current best view is that effective dose is individual. Patients differ in sensitivity, metabolism, symptom profile, and underlying biology. A rigid one-dose-for-all approach is not supported by the newer observational data.
| Dose range / approach | Typical use | Practical note |
|---|---|---|
| 0.1–0.25 mg | Very sensitive patients | Often useful in MCAS, ME/CFS and autonomic reactivity |
| 0.5–1 mg | Common starting range | Suitable for many standard LDN starts |
| 0.5–4.5 mg | Typical clinical range | Most commonly discussed in clinical practice |
| Above 4.5 mg | Selected patients | Occasionally used when partial response plateaus |
| Ultra-low-dose (micrograms) | Specialist / experimental opioid modulation | Not the same as routine stand-alone LDN prescribing |
Pharmacokinetics, Receptor Blockade and Practical Implications for Opioid Use
Oral naltrexone has a relatively short plasma half-life, but its clinical effects — particularly opioid receptor blockade — persist significantly longer than plasma concentrations alone would suggest.
| Pharmacokinetic data | Value |
|---|---|
| Naltrexone elimination half-life | Approximately 4–5 hours |
| Active metabolite (6-beta-naltrexol) half-life | Approximately 13 hours |
PET imaging studies using standard 50 mg doses suggest that central mu-opioid receptor blockade persists far longer than the plasma half-life alone would imply, with clinically meaningful opioid attenuation for 24–72 hours after a dose in some individuals. However, these pharmacodynamic data come mainly from standard-dose naltrexone, not LDN specifically.
LDN is used in much lower doses, so the blockade is likely to be shorter than with standard 50 mg dosing. However, there are no robust PET studies specifically evaluating receptor occupancy at typical LDN doses. In practice, most perioperative guidance extrapolates from standard-dose experience and from real-world analgesia problems rather than relying on half-life calculations alone.
For planned opioid use, such as elective surgery or a short course of strong analgesia, a practical approach is usually to withhold LDN for 48–72 hours beforehand. If opioids are needed urgently, they may still be used, but reduced or less predictable efficacy should be anticipated and multimodal non-opioid analgesia should be optimised where possible.
Key clinical message: Clinical decisions around opioids and surgery should be based on functional receptor recovery and analgesic reliability, not plasma half-life alone.
Suggested LDN Starting and Titration Schedules
A practical way to start is low and build slowly. Traditional fact sheets describe beginning at around 0.5 mg to 1.5 mg depending on the indication and increasing every two weeks as tolerated. In more sensitive patients, especially those with MCAS, ME/CFS or marked autonomic reactivity, even lower starting doses may be better.
Low and Slow Dosing Pearl
In sensitive patients — particularly those with ME/CFS, MCAS, EDS, POTS or marked autonomic reactivity — starting too high can trigger a flare. A lower starting dose and slower titration often improve tolerability and long-term success.
Standard starting schedule
A typical adult starting plan might be 0.5 mg to 1 mg nightly, increasing gradually every 1 to 2 weeks.
Slow titration schedule for sensitive patients
For very reactive patients, starting at 0.1 mg to 0.25 mg and titrating much more slowly can make the difference between success and an early flare. The LDN Research Trust page on EDS/POTS/MCAS strongly reflects this practical “start very low, increase very slowly” approach.
Ultra-low-dose schedule
This belongs more to specialist or research-style opioid modulation than routine LDN prescribing.
Higher-dose pathway when clinically appropriate
If a patient improves partially but plateaus, some clinicians cautiously titrate above 4.5 mg, but this should be individualized rather than routine.
Morning versus evening dosing
Night dosing is traditional, but some patients sleep poorly or develop vivid dreams. In those cases morning dosing is a sensible alternative and may improve tolerability.
| Patient group | Typical starting dose | Titration approach | Practical notes |
|---|---|---|---|
| Standard adult start | 0.5–1 mg nightly | Increase every 1–2 weeks | Common starting approach in routine practice |
| Sensitive patients | 0.1–0.25 mg | Much slower titration | Helps reduce early flares and poor tolerability |
| ME/CFS / Long COVID | Often lower than standard | Slow, patient titration | Often better tolerated with cautious escalation |
| MCAS / EDS / autonomic reactivity | 0.1 mg or 0.25 mg | Very low, very slow | Formulation and excipients may matter |
| Partial response plateau | Above 4.5 mg in selected cases | Individualised only | Not a routine default pathway |
How Long Should an LDN Trial Last Before Judging Response?
LDN is not a medication that should be judged after a few days. In fibromyalgia reviews, a 3-month trial is considered reasonable, and in long COVID studies improvements were assessed over 8 to 12 weeks. For many patients, especially those with complex fatigue syndromes, a proper trial means patient, gradual titration and adequate time at a tolerated dose.
What Improvement Might Patients Notice First?
Pain
Pain reduction is often one of the earliest meaningful changes in responders, especially in fibromyalgia and centralised pain states.
Energy
In ME/CFS and long COVID, some patients report a later, more gradual improvement in energy or recovery capacity rather than an immediate lift.
Sleep
Sleep can improve, but some patients initially notice vivid dreams or insomnia before things settle.
Brain fog / concentration
Brain fog can improve in some long COVID and ME/CFS patients, though this tends to be gradual rather than dramatic.
Flare patterns and delayed response
Response is often non-linear. Some patients feel worse before better, particularly if the dose is pushed too fast. That is one reason slow titration matters so much in sensitive groups.
| Timeframe | What patients may notice | Practical interpretation |
|---|---|---|
| First 1–2 weeks | Sleep changes, vivid dreams, possible early flare | Not necessarily treatment failure; often part of early adjustment |
| 2–6 weeks | Pain may begin to improve in responders | Pain is often one of the earliest clearer changes |
| 6–12 weeks | Energy, recovery capacity, brain fog may start to shift | Often requires patience and time at a tolerated dose |
| Beyond 12 weeks | More stable pattern of benefit may emerge | Especially relevant in long COVID and ME/CFS |
Side Effects and Safety of LDN
LDN is generally well tolerated, and the side-effect burden is usually modest compared with many chronic pain medications. That said, it is not side-effect free and patients should be counselled honestly.
Common side effects
The most common issues are vivid dreams, insomnia, headache and occasional gastrointestinal upset.
Vivid dreams, sleep disturbance and headache
These are repeatedly described in both research and practical guidance. Vivid dreams may begin quickly and often settle with time.
Gastrointestinal effects
Nausea, diarrhoea, constipation or stomach upset can occur, though they are usually uncommon and mild.
Mood effects, anxiety and palpitations
Some clinicians have reported anxiety or tachycardia anecdotally. In highly sensitive patients, these can be signals that the starting dose was too high.
Hepatic considerations and general tolerability
Serious liver toxicity is not a major issue at LDN doses, but caution is reasonable in significant liver disease.
| Side effect | How it may present | Practical management |
|---|---|---|
| Vivid dreams | Unusual or intense dreaming early in treatment | Often settles with time; consider morning dosing if needed |
| Insomnia / sleep disruption | Difficulty falling or staying asleep | Consider dose reduction, slower titration or morning dosing |
| Headache | Early treatment headache | Often transient; slow titration may help |
| Gastrointestinal upset | Nausea, diarrhoea, constipation, stomach upset | Usually mild; review formulation if sensitive |
| Anxiety / palpitations | Sense of activation, anxiety or tachycardia | May indicate starting dose was too high |
| Temporary flare | Early worsening before improvement | Slow down titration; reassess rather than stopping too early |
LDN and Opioids: Why They Should Generally Not Be Used Together
Important Safety Point
Standard LDN should generally not be used alongside regular opioid therapy. If opioid analgesia becomes necessary, LDN is often paused temporarily and restarted once opioid treatment is no longer needed.
This is one of the most important practical sections in the whole article. Standard LDN is generally avoided with ongoing opioid therapy because naltrexone blocks opioid receptors for several hours and may reduce opioid effectiveness or complicate treatment. Practical fact sheets advise that patients should not start LDN until opioid agonists are out of the system.
Why concomitant opioid use is problematic
Because the drug is an opioid antagonist, it can blunt the expected effect of opioid analgesics.
Risk of reduced opioid effect, withdrawal-type symptoms or mixed response
Frank withdrawal seems uncommon at low doses, but reduced analgesia, gastrointestinal side effects, or an unpleasant mixed response are still concerns.
Special caution for patients taking regular opioid analgesics
Patients taking regular opioid analgesics such as tramadol, codeine, morphine, oxycodone or similar medications require particularly careful assessment before low-dose naltrexone (LDN) is considered.
The key issue is pharmacological. Naltrexone is an opioid receptor antagonist, and even at low doses it may interfere with opioid receptor signalling for several hours after administration. In practical terms, this can result in reduced analgesic efficacy of opioids, unpredictable pain control, or in some cases an unpleasant mixed clinical picture including agitation, nausea or withdrawal-type symptoms. Although frank withdrawal is uncommon with standard LDN dosing, the interaction is clinically significant and should not be overlooked.
For patients using intermittent opioids, LDN may be introduced once opioid use has ceased and the medication has cleared from the system. However, in patients taking regular daily opioids, particularly where physiological dependence has developed, LDN should generally not be started immediately.
A more appropriate approach is a structured transition. This begins with careful review of the indication for ongoing opioid therapy, followed by gradual reduction where feasible and optimisation of non-opioid pain management strategies. In dependent patients, a more formal opioid weaning process may be required before LDN can be introduced safely. In practical terms, this often means reducing and stopping regular opioid therapy first, then starting LDN only once the patient is no longer taking opioid agonists.
This stepwise approach is both safer and more likely to produce a meaningful therapeutic response. It also allows the clinician to judge more clearly whether LDN is helping, rather than introducing it into an unstable analgesic regimen.
Important distinction between LDN and ultra-low-dose naltrexone used experimentally with opioids
Ultra-low-dose naltrexone used experimentally to modify opioid signalling is not the same as routine LDN prescribing and should not be conflated.
What If a Patient on LDN Needs Opioids?
Patients taking LDN may still require opioid analgesia in certain situations, including acute injury, severe pain exacerbations, emergency care, or perioperative management. The presence of LDN does not permanently prevent the use of opioids, but it does require a more thoughtful and structured approach.
Acute pain situations
If a patient taking LDN develops acute severe pain, such as after a fracture, renal colic, major dental pain, or another emergency presentation, opioid analgesia may still be appropriate. However, because LDN may reduce opioid responsiveness for a period after each dose, a practical approach is to withhold LDN temporarily if opioids are likely to be required. This helps avoid reduced or unpredictable opioid effect and may reduce the risk of unnecessary opioid dose escalation.
Severe flare-ups
In severe pain flares, management should be individualised. If symptoms can be controlled with non-opioid strategies, LDN can usually be continued. If escalation to opioid therapy is required, it is often more practical to pause LDN temporarily, manage the flare appropriately, and then resume LDN once opioid use is no longer necessary. This is more useful clinically than a simple instruction never to combine the two, because it gives the patient and clinician a workable plan.
Emergency care considerations
Patients should be advised to inform all healthcare professionals — including emergency clinicians, dentists, surgeons and anaesthetists — that they are taking LDN. Because LDN is often prescribed as a compounded medication, patients do not always realise it is relevant in acute settings. In reality, this information may directly influence analgesic planning.
Practical communication advice for patients and clinicians
A helpful and simple message is this: if strong opioid pain relief is likely to be needed, LDN will usually be paused temporarily and restarted afterwards. Clear communication between the patient, prescriber, surgeon and anaesthetist is particularly important in planned care.
Restarting LDN after opioid use
Once opioid analgesia is no longer required and has cleared from the system, LDN can usually be restarted safely. If the interruption was brief and the patient had previously tolerated LDN well, some clinicians will restart the previous dose. In more sensitive patients — especially those with ME/CFS, long COVID, MCAS or marked autonomic reactivity — it may be more sensible to restart at a slightly lower dose and titrate back up more gradually.
| Situation | What to do with LDN | Key caution |
|---|---|---|
| Acute severe pain | Often pause temporarily | Opioid response may be reduced if LDN taken recently |
| Severe flare-up | Individualise | Do not casually layer LDN and opioids without a plan |
| Emergency care | Inform treating clinicians | Compounded medicines are still clinically important |
| Restart after opioids | Restart once opioids no longer required | Sensitive patients may need lower restart dose |
LDN in the Perioperative Period
Why this matters before surgery or procedures
Perioperative care matters because postoperative opioid requirements are common, and opioid receptor antagonism may complicate analgesia.
Potential interaction with perioperative opioid analgesia
The core issue is the same as above: reduced opioid responsiveness at a time when effective pain control is important.
When LDN may need to be stopped before surgery
For surgery or procedures where opioid analgesia is expected, LDN will often need to be stopped in advance. Although oral naltrexone has a relatively short plasma half-life, the clinical effect on opioid receptor availability lasts longer than plasma values alone would suggest.
From a pharmacokinetic perspective, oral naltrexone has a parent half-life of approximately 4 to 5 hours, while its active metabolite, 6-beta-naltrexol, has a longer half-life of approximately 13 hours. However, pharmacodynamic effects extend beyond these figures. PET studies using standard 50 mg doses of naltrexone suggest that central mu-opioid receptor blockade persists far longer than the plasma half-life alone would imply, with a blockade half-time of around 72 hours in some studies and clinically relevant receptor occupancy potentially lasting several days.
LDN is used at much lower doses, so the duration of receptor blockade is likely to be shorter than with standard 50 mg dosing. However, there are no robust PET data specifically defining receptor occupancy at typical LDN doses. For this reason, most clinical guidance extrapolates from standard-dose experience and from real-world analgesia problems rather than relying on half-life calculations alone.
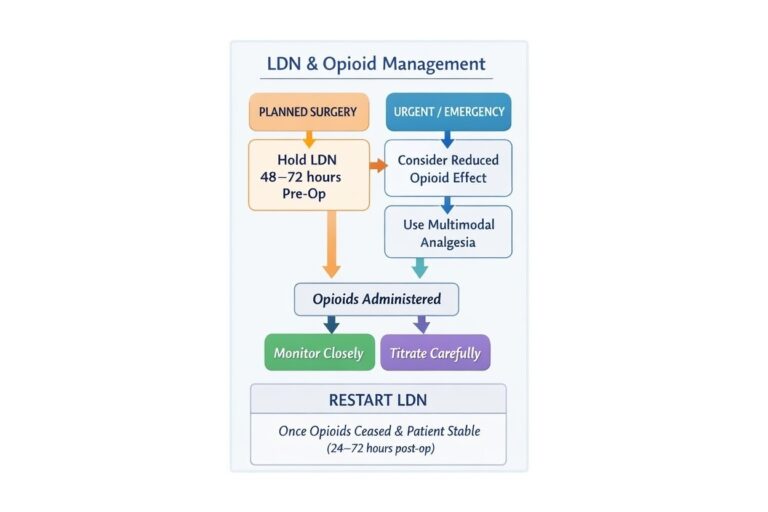
A practical and defensible approach is therefore to stop LDN 48 to 72 hours before planned procedures where opioid analgesia is likely to be needed, assuming normal renal and hepatic function. The shorter end of that range may be reasonable for minor procedures or lower expected opioid requirements, while the longer end is often preferred where major surgery or substantial postoperative pain is anticipated.
The key point is that perioperative planning should be based on achieving reliable opioid analgesia, not simply on the parent-drug half-life. Minor procedures managed without opioids may be different, but for more significant surgery it is sensible for the patient, surgeon, anaesthetist and prescriber to agree the plan in advance.
Restarting LDN after surgery
LDN can usually be restarted once opioid analgesia is no longer required and the patient is clinically stable. Restarting too early may blunt opioid effect and complicate pain control. In a patient who has been off LDN only briefly and previously tolerated it well, the prior dose may often be resumed. In more sensitive patients, it may be reasonable to restart at a slightly lower dose and titrate back upwards again.
Why perioperative planning should be individualised
Minor procedures managed without opioids may be different from major surgery. Some patients will need only simple analgesia, while others may require substantial perioperative opioid support. This is one area where blanket advice is less helpful than explicit communication between the patient, prescriber, surgeon and anaesthetic team. The most important practical principle is to ensure that LDN does not compromise reliable postoperative pain control.
| Scenario | Practical approach | Why it matters |
|---|---|---|
| Minor procedure without opioids | May be individualised | Not every procedure requires the same washout plan |
| Elective surgery with likely opioid use | Often stop LDN 48–72 hours beforehand | Helps avoid reduced opioid responsiveness |
| Major surgery | Use longer end of stop window and coordinate plan | Reliable postoperative analgesia is critical |
| Restart after surgery | Restart once opioids no longer required | Avoid blunting opioid effect too early |
When LDN May Not Be Suitable
Current opioid use
This is the clearest practical contraindication.
Recent opioid dependence treatment
Patients with recent opioid treatment or dependence need specialist input.
Pregnancy / breastfeeding considerations
Evidence is limited, so routine use should be cautious.
Significant liver disease or complex prescribing situations
These require individual review rather than routine prescribing.
Need for specialist review
Complex multisystem patients, especially those with severe autonomic instability or mast cell disease, benefit from careful supervision rather than generic prescribing.
What the Evidence Really Shows: Promise, Limitations and Unanswered Questions
Where evidence is strongest
Fibromyalgia and centralised chronic pain have the best developed evidence base, though still modest.
Where evidence is emerging
Long COVID and ME/CFS are promising but still developing areas.
Where evidence is still limited or indirect
PTSD, MCAS and autonomic syndromes remain less directly studied.
Why more high-quality trials are needed
Small sample sizes, off-label use, variable dosing and heterogeneous patients make it hard to draw firm conclusions. The field clearly needs better trials, not just enthusiasm.
Practical Clinical Take-Home Points
LDN is best thought of as a trial of neuroimmune modulation in carefully selected patients rather than as a conventional painkiller. It is particularly relevant where chronic pain overlaps with fatigue, poor sleep, hypersensitivity, brain fog and central sensitisation. Dosing should be individualized. Sensitive patients often need much lower starting doses and slower titration than standard protocols imply. Above all, opioid compatibility must be considered from the outset.
Conclusion: Where LDN May Fit in Modern Pain and Post-Viral Care
LDN occupies an interesting and increasingly important space in modern pain medicine. It is not a cure, not a mainstream first-line drug, and not equally supported across all diagnoses. But it does offer something different: a plausible mechanism-based option for patients whose symptoms sit at the interface of pain, fatigue, inflammation, sensitisation and dysautonomia. In that sense, it fits especially well into contemporary Pain Spa-style practice, where we are often treating not just pain intensity, but the wider biology of amplification, overprotection and poor system regulation.
Frequently Asked Questions (FAQ)
1. Is LDN a painkiller?
Not in the traditional sense. It is better understood as a modulator of neuroimmune and pain-processing pathways rather than a direct analgesic.
2. What is the usual dose?
Many patients end up somewhere between 0.5 mg and 4.5 mg daily, but there is no single correct dose for everyone. Some do better lower, some higher.
3. How do you start it?
Usually low and slow. A common strategy is to start at 0.5 mg to 1 mg, but highly sensitive patients may need 0.1 mg to 0.25 mg.
4. How long should I try it for?
A fair trial is usually at least 8 to 12 weeks, often longer in post-viral fatigue syndromes.
5. Can it make me feel worse at first?
Yes. Some patients notice sleep disturbance, vivid dreams, headache, agitation or a flare if the starting dose is too high or increased too quickly.
6. Can it help brain fog?
It may, particularly in long COVID or ME/CFS, but improvement is usually gradual rather than immediate.
7. Can I take it with opioids?
Generally, standard LDN should not be combined casually with regular opioid therapy. If opioids are required for acute pain, LDN is often paused temporarily and restarted once opioid treatment has finished.
8. What if I need surgery?
Tell your surgeon and anaesthetist well in advance. If opioids are likely to be needed, LDN is often stopped 48–72 hours beforehand and restarted once opioid analgesia is no longer required.
9. Should I take it in the morning or at night?
Night is traditional, but morning dosing is reasonable if you develop insomnia or vivid dreams.
10. Can people with MCAS or EDS take LDN?
Yes, but usually with much more cautious dosing and careful choice of formulation.
11. Is it safe long term?
So far it appears generally well tolerated, but long-term high-quality safety data in chronic low-dose use are still limited.
12. Does failure at 4.5 mg mean LDN does not work for me?
Not necessarily. Some patients respond only at lower or higher doses, so a fixed-dose failure is not always a true treatment failure.
13. Can it help fibromyalgia, long COVID and ME/CFS if they overlap?
Potentially yes. Those conditions overlap substantially, and that overlap is part of the rationale for trying LDN.
14. Is LDN available as a standard NHS tablet?
Usually no. It is commonly prescribed as a compounded preparation.
15. What is the best way to think about LDN overall?
As a carefully supervised, individualized trial in the right patient, not as a guaranteed answer and not as a replacement for broader pain, autonomic, rehabilitation or psychological care.
Selected References
Toljan K, Vrooman B. Low-Dose Naltrexone (LDN)—Review of Therapeutic Utilization. Med Sci. 2018.
If you want to consider taking LDN, please contact Pain Spa at clinic@painspa.co.uk or visit www.painspa.co.uk for further information.